CHEMISTRY UGC’s Draft Model Syllabi) (PROPOSED SCHEME)
UGC CBCSTDC COURSE CURRICULUMandSYLLABI(As per UGC’s Draft ModelSyllabi)(PROPOSED SCHEME)CHEMISTRYUGC’s Draft Model Syllabi)(PROPOSED SCHEME)Submitted by:Dr. Pradip C. PaulHead, Department of ChemistryChairman, BUGS-ChemistryAssam University: Silchar.Adopted in the meeting of the BUGS-Chemistry dated April 20, 2017.Modified as per CBCS Syllabus Structure Preparation Committee Meeting,March 28, 2017
Contents1. CBCS UG - Course Curriculum – Basic Structure2. CBCS UG – SYLLABI for CHEMISTRY (HONOURS)a) Semester-Wise Course Structure for Choice Based Credit System In B. Sc. Honours(Chemisry)b) Core Courses for Chemistry (Honours) with Course Code and Course Namec) Discipline Specific Elective (DSE) Courses for Chemistry Honoursd) Skill Enhancement Courses (SEC)e) Semester wise list of Chemistry Generic Elective papers for the students taking Honours inother disciplines3. CBCS –Syllabi for B. Sc. with Chemistrya) Semester-Wise Course Structure for Choice Based Credit System in B. Sc. with Chemistryb) Semester wise list of Chemistry papers to be studied as Discipline Specific Core (DSC)Courses by students of BSc with Chemistryc) Discipline Specific Elective (DSE) Coursesd) Skill Enhancement Courses (SEC)4. Detailed Syllabia) Core Coursesb) Discipline Specific Elective (DSE) Coursesc) Skill Enhancement Courses (SEC)Important Note:Course content, Question Papers and Schedule of Examination shall be the same for DSC[Core for BSc with Chemistry] and GE [Hons with other Discipline] in accordance withsections 2.3, 2.1 and 3.2 of UGC’s Instructional Template for facilitating implementation ofCBCS.Reference:UGC’s Instructional Template for Facilitating Implementation of CBCS2.3 P.S.:A core course offered in a discipline/subject may be treated as an elective byother discipline/subject and vice versa and such electives may also bereferred to as Generic Elective (GE).2.1Discipline Specific Elective (DSE) (4 for Honours and 2 for Regular courses)3.2Skill Enhancement Courses (SEC) (2 for Honours and 4 for Regular courses).
CBCS UG - COURSE CURRICULUM (Basic structure)B.Sc. (Honours)CHEMISTRY (HONOURS)CORE COURSE (C)CHEMISTRYCORETheory PracticalPapersxCreditsTotal Credits14x4 284Chemistry Theory Practical4x4 224Theory Practical4x4 2ELECTIVE COURSE (DSE and GE)DSEGEor24orTheory Tutorial4x5 1GE Courses are Computer Science, Mathematics, Physics (any one).Ability Enhancement CoursesAbility Enhancement Compulsory Courses (AECC)1) English/MIL Communication2) Environmental Science2x408Skill Enhancement Courses (SEC)2x408Total Credits 148CBCS UG - Course CurriculumB. Sc. with ChemistryDiscipline Specific Core (DSC) CoursesDSCPapersxCredits444xxx4 24 24 24x5 1222xxx4 24 24 22x5 14x4162x408DSC 1: Chemistry Theory PracticalDSC 2: other subject Theory PracticalDSC 3: other subject Theory PracticalorDSC 3: other subject Theory TutorialTotal Credits72Discipline Specific Elective (DSE) CoursesDSEDSE 1: Chemistry Theory PracticalDSE 2: other subject Theory PracticalDSE 3: other subject Theory PracticalorDSC E: other subject Theory TutorialSkill Enhancement Courses (SEC)Ability Enhancement Compulsory Courses(AECC)361) English/MIL Communication2) Environmental ScienceTotal Credits Each credit is equivalent to 1 hour of activity per week132
CBCSTDC - SYLLABICHEMISTRY(HONOURS)
SEMESTER-WISE COURSE STRUCTURE FOR CHOICE BASED CREDIT SYSTEM INB. Sc. Honours (CHEMISRY)CORE COURSE(14 papers)IElective: DisciplineSpecific DSE(4 papers)CHEMISTRY -C-301Elective:Generic(GE)4 papers(To be taken fromother discipline)GE-1EnvironmentalScienceCHEMISTRY -C-201CHEMISTRY -C-202IIIAbility EnhancementElective Course(AEEC)(Skill Based)(2 papers)CHEMISTRY-C-101CHEMISTRY -C-102IIAbility EnhancementCompulsory Course(AECC)(2 1GE-3CHEMISRY-SEC-401GE-4CHEMISTRY -C-302CHEMISTRY -C-303IV CHEMISTRY -C-401CHEMISTRY -C-402CHEMISTRY -C-403V CHEMISTRY -C-501CHEMISRY-DSE-501CHEMISTRY -C-502CHEMISRY-DSE -502VI CHEMISTRY -C-601CHEMISRY-DSE -601CHEMISTRY -C-602CHEMISRY-DSE -602
Core Courses for Chemistry (Honours) with Course Code and Course NameSEMESTERICourse 201-LABCHEMISTRY-C-202Course NameInorganic Chemistry –IAtomic Structure and Chemical BondingPracticalPhysical Chemistry –IStates of Matter and Ionic EquilibriumPracticalOrganic Chemistry –IHydrocarbons and StereochemistryPracticalPhysical Chemistry 0Chemical Thermodynamics and its STRY-C-602CHEMISTRY-C-602-LABPracticalInorganic Chemistry –IIs- & p-block Elements and MetallurgyPracticalOrganic Chemistry –IIHalogen & Oxygen Containing FunctionalGroupsPracticalPhysical Chemistry –IIIPhase Equilibria and Chemical KineticsPracticalInorganic Chemistry –IIICoordination Chemistry and its ApplicationsPracticalOrganic Chemistry –IIIHeterocyclic ChemistryPracticalPhysical Chemistry –IVElectrochemistryPracticalOrganic Chemistry –IVBiomoleculesPracticalPhysical Chemistry –VQuantum Chemistry and SpectroscopyPracticalInorganic Chemistry –IVOrganometallic ChemistryPracticalOrganic Chemistry –VSpectroscopy, Dyes and PolymersPractical
Discipline Specific Elective (DSE) Courses forChemistry HonoursSEMESTER COURSE No.Couse NameCreditMarksCHEMISTRY-DSE-501Analytical Methods in STRY-DSE-502Green STRY-DSE-601Inorganic Materials of cal230CHEMISTRY-DSE-602Dissertation (Project Work)6100VVISkill Enhancement Courses (SEC)(FOR CHEMISTRY HONOURS)IIICHEMISTRY-SEC-301Analytical Clinical Biochemistry470IVCHEMISTRY –SEC-401Fuel Chemistry470
Semester wise list of Chemistry Generic Elective (GE) papers for the studentstaking Honours in other disciplinesSEMESTERIIIIIICOURSE No.Course NameCHEMISTRY-GE-101Atomic Structure, Bonding, General OrganicChemistry and Aliphatic -GE-201Chemical Energetics, Equilibria andFunctional Group Organic GE-301Solutions, Phase Equilibrium,Conductance, Electrochemistry andFunctional Group Organic RY-GE-401Transition metals, Coordination Chemistry,States of Matter and Chemical 0230470230470230470230
CBCSTDC - SYLLABIB. Sc. withCHEMISTRY
SEMESTER-WISE COURSE STRUCTURE FOR CHOICE BASED CREDIT SYSTEM INB. Sc. with ChemistryDISCIPLINE SPECIFICCORECOURSE (DSC)(12 papers)CHEMISTRY-DSC-101IDSC- 2 AAbility EnhancementSkillCompulsoryEnhancement Course (SEC)Course(AECC)(4 papers)(2 papers)Discipline SpecificElective (DSE)(6 papers)EnvironmentalScienceDSC- 3 ACHEMISTRY-DSC-201IIEnglish/MILCommunicationDSC- 2 BDSC- 3 BCHEMISTRY-DSC-301IIICHEMISTRY-SEC-301DSC- 2 CDSC- 3 CCHEMISTRY-DSC-401IVCHEMISTRY-SEC-401DSC- 2 DDSC- 3 DCHEMISTRY-SEC-501CHEMISTRY-DSE-501DSE-2 AVDSE-3 ACHEMISTRY-SEC-601VICHEMISTRY-DSE-601DSE-2 BDSE-3 B
CBCS: B. Sc. with ChemistrySemester wise list of Chemistry papers to be studied as Discipline Specific Core (DSC) Courses bystudents of BSc with Chemistry (Regular)COURSE No.CreditMarksSEMESTERCourse NameCHEMISTRY-DSC-101470Atomic Structure, Bonding, General Organic Chemistry andIAliphatic STRY-DSC-201470Chemical Energetics, Equilibria andFunctional Group Organic MISTRY-DSC-30170Solutions, Phase Equilibrium, Conductance, Electrochemistryand Functional Group Organic MISTRY-DSC-401Transition metals, Coordination Chemistry,States of Matter and Chemical 230470230470470470470Discipline Specific Elective (DSE) CoursesVVICHEMISTRY-DSE-501Analytical Methods in SE-601Inorganic Materials of Industrial ImportanceCHEMISTRY-DSE-601-LABPracticalSkill Enhancement Courses (SEC)IIIIVVVICHEMISTRY-SEC-301Analytical Clinical BiochemistryCHEMISTRY-SEC-401Fuel ChemistryCHEMISTRY-SEC-501Chemistry of Cosmetics and PerfumesCHEMISTRY-SEC-601Pesticide ChemistryAssam University: Silchar.CBCS UG CHEMISTRYPage 7 of 99
Detailed SyllabiAssam University: Silchar.CBCS UG CHEMISTRYPage 8 of 99
CBCS: B. Sc. (Honours) with CHEMISTRYCORE COURSECHEMISTRY(Honours)(1st Semester)Course No.:CHEMISTRY-C-101(Inorganic Chemistry – I)Atomic Structure & Chemical BondingContact Hours: 60; Credits: 04Full Marks 70[End Semester Exam (50) Internal Assessment (20)]Pass Marks 28 [End Semester Exam (20) Internal Assessment(08)](In the End-semester examination, two questions of 10 marks will be set from each unit,one needs to be answered from each unit)Objective of thiscourse is to emphasize on Atomic structure, bonding and periodicityUnit 1: Atomic StructureBohr’s theory, its limitations and atomic spectrum of hydrogen atom. Wave mechanics: deBroglie equation, Heisenberg’s Uncertainty Principle and its significance, Schrödinger’swave equation, significance of and 2. Quantum numbers and their significance.Normalized and orthogonal wave functions. Sign of wave functions. Radial and angular wavefunctions for hydrogen atom. Radial and angular distribution curves. Shapes of s, p, d and forbitals. Contour boundary and probability diagrams.Pauli’s Exclusion Principle, Hund’s rule of maximum multiplicity, Aufbau’s principle and itslimitations.Unit 2: Periodicity of Elementss , p , d , f block elements, the long form of periodic table. Detailed discussion of thefollowing properties of the elements, with reference to s &p block.(a)Effective nuclear charge, shielding or screening effect, Slater rules, variation ofeffective nuclear charge in periodic table.(b)Atomic radii (van der Waals)(c)Ionic and crystal radii.(d)Covalent radii (octahedral and tetrahedral)(e)Ionization enthalpy, Successive ionization enthalpies and factors affecting ionizationenergy. Applications of ionization enthalpy.(f)Electron gain enthalpy, trends of electron gain enthalpy.(g)Electronegativity, Pauling’s/ Mulliken’s/ Allred Rachow’s electronegativity scales.Variation of electronegativity with bond order, partial charge, hybridization, groupelectronegativity.Assam University: Silchar.CBCS UG CHEMISTRYPage 9 of 99
Unit 3: Chemical Bonding I(i) Ionic bond: General characteristics, types of ions, size effects, radius ratio rule and itslimitations. Packing of ions in crystals. Madelung constant, Born-Haber cycle and itsapplication, Solvation energy.(ii) Covalent bond: Lewis structure, Valence Bond theory (Heitler-London approach).Energetics of hybridization, equivalent and non-equivalent hybrid orbitals. Resonance andresonance energy, Molecular orbital theory. Molecular orbital diagrams of diatomic andsimple polyatomic molecules N2, O2, C2, B2, F2, CO, NO, and their ions; HCl, BeF2, CO2,(idea of s-p mixing and orbital interaction to be given). Formal charge, Valence shell electronpair repulsion theory (VSEPR), shapes of simple molecules and ions containing lone pairsand bond pairs of electrons, multiple bonding (σ and π bond approach) and bond lengths.Unit 4: Chemical Bonding II(i)Ionic character in covalent compounds: Bond moment and dipole moment. Percentageionic character from dipole moment and electronegativity difference.(ii) Covalent character in ionic compounds, polarizing power and polarizability. Fajan’srules and consequences of polarization.(iii) Metallic Bond: Qualitative idea of valence bond and band theories. Semiconductors andinsulators, defects in solids.(iv) Weak Chemical Forces: van der Waals forces, ion-dipole forces, dipole-dipoleinteractions, induced dipole interactions, Instantaneous dipole-induced dipoleinteractions. Hydrogen bonding (theories of hydrogen bonding, valence bondtreatment).Unit 5: Oxidation-ReductionRedox reactions, balancing Redox equations by ion-electron method Standard ElectrodePotential and its application to inorganic reactions, concept of formal Electrode PotentialPrinciples involved in volumetric analysis: Fe estimation by standard KMnO4 solution, andK2Cr2O7 solution.Reference Books: Lee, J. D. Concise Inorganic Chemistry, ELBS, 1991. Douglas, B. E. and Mc Daniel, D. H., Concepts & Models of Inorganic Chemistry,Oxford, 1970 Atkins, P. W. & Paula, J. Physical Chemistry, Oxford Press, 2006. Day, M. C. and Selbin, J. Theoretical Inorganic Chemistry, ACS Publications 1962.Assam University: Silchar.CBCS UG CHEMISTRYPage 10 of 99
CBCS: B. Sc. (Honours) with CHEMISTRYCORE COURSECHEMISTRY LAB(Honours)(1st Semester)Course No.:CHEMISTRY-C-101-LAB(Inorganic Chemistry – I)PracticalContact Hours: 60; Credits: 02Full Marks 30Pass Marks 1225Examination Time: 12 hours (spread over two days)During examination, Two experiments(one from each of the Part I and Part II), to be assignedto each student by drawing lots.1.2.Part I: Titrimetric Analysis047 marks(i)Calibration and use of apparatus(ii)Preparation of solutions of different Molarity/Normality of titrantsPart II: Oxidation-Reduction Titrimetry2014 marks(i)Estimation of Fe (II) and oxalic acid using standardized KMnO4 solution.(ii)Estimation of Fe (II) with K2Cr2O7solution.3.Viva – voce032 marks4.Regularity in maintenance of Lab Notebook03 2 marks5.Attendance03 5 marksReference Books: Vogel, A. I., A Textbook of Quantitative Inorganic Analysis, ELBS. Nad, A.K., Mahapatra, B., Ghoshal, A., An Advanced Course in Practical Chemistry,New Central Book Agency (P) Ltd., Kolkata, India. Das, Subhas C, Advanced Practical Chemistry for 3-Year Honours Course.Assam University: Silchar.CBCS UG CHEMISTRYPage 11 of 99
CBCS: B. Sc. (Honours) with CHEMISTRYCORE COURSECHEMISTRY(Honours)(1st Semester)Course No.:CHEMISTRY-C-102(Physical Chemistry – I)States of Matter & Ionic EquilibriumContact Hours: 60; Credits: 04Full Marks 70[End Semester Exam (50) Internal Assessment (20)]Pass Marks 28 [End Semester Exam (20) Internal Assessment(08)](In the End-semester examination, two questions of 10 marks will be set from each unit,one needs to be answered from each unit)Objective of this course is to emphasize on different states of matter and their mathematicaltreatment.Unit 1: Gaseous State IKinetic molecular model of a gas: postulates and derivation of the kinetic gas equation;collision frequency; collision diameter; mean free path and viscosity of gases, including theirtemperature and pressure dependence, relation between mean free path and coefficient ofviscosity; variation of viscosity with temperature and pressure.Maxwell distribution and its use in evaluating molecular velocities (average, root meansquare and most probable) and average kinetic energy, law of equipartitions of energy anddegrees of freedom.Unit 2: Gaseous State IIBehaviour of real gases: Deviations from ideal gas behaviour, compressibility factor, Z, andits variation with pressure for different gases. Causes of deviation from ideal behaviour. vander Waals equation of state, its derivation and application in explaining real gas behaviour,mention of other equations of state (Berthelot, Dietrici); virial equation of state. Isotherms ofreal gases and their comparison with van der Waals isotherms, continuity of states, criticalstate, relation between critical constants and van der Waals constants, law of correspondingstates.Unit 3: Liquid StateQualitative treatment of the structure of the liquid state; Radial distribution function; physicalproperties of liquids; vapour pressure, surface tension and coefficient of viscosity, and theirdetermination. Effect of addition of various solutes on surface tension and viscosity.Explanation of cleansing action of detergents. Temperature variation of viscosity of liquidsand comparison with that of gases.Assam University: Silchar.CBCS UG CHEMISTRYPage 12 of 99
Unit 4: Solid StateNature of the solid state, law of constancy of interfacial angles, law of rational indices, Millerindices, elementary ideas of symmetry, symmetry elements and symmetry operations,qualitative idea of point groups, seven crystal systems and fourteen Bravais lattices; X-raydiffraction, Bragg’s law. Defects in crystals. Glasses and liquid crystals.Unit 5: Ionic EquilibriaStrong, moderate and weak electrolytes, degree of ionization, factors affecting degree ofionization, ionization constant and ionic product of water. Ionization of weak acids and bases,pH scale, common ion effect; dissociation constants of mono-, di-and triprotic acids.Salt hydrolysis-calculation of hydrolysis constant, degree of hydrolysis and pH for differentsalts. Buffer solutions; derivation of Henderson equation and its applications; buffer capacity,buffer range, buffer action and applications of buffers in analytical chemistry andbiochemical processes in the human body.Solubility and solubility product of sparingly soluble salts – applications of solubility productprinciple. Qualitative treatment of acid – base titration curves (calculation of pH at variousstages). Theory of acid–base indicators; selection of indicators and their limitations.Reference Books: Atkins, P. W. & Paula, J. de Atkin’s Physical Chemistry Ed., Oxford University Press 13(2006). Ball, D. W. Physical Chemistry Thomson Press, India (2007). Castellan, G. W. Physical Chemistry 4th Ed. Narosa (2004). Mortimer, R. G. Physical Chemistry 3 rd Ed. Elsevier: NOIDA, UP (2009).Assam University: Silchar.CBCS UG CHEMISTRYPage 13 of 99
CBCS: B. Sc. (Honours) with CHEMISTRYCORE COURSECHEMISTRY LAB(Honours)(1st Semester)Course No.:CHEMISTRY-C-102-LAB(Physical Chemistry – I)PracticalContact Hours: 60; Credits: 02Full Marks 30Pass Marks 1225Examination Time: 12 hours (spread over two days)During examination, Two experiments (one from each of the Part I and Part II), to beassigned to each student by drawing lots.1. Part I1610 marksa) Determination of transition temperature of the given substance by thermometric method(e.g., MgSO4/MnCl2/Na2SO4.10H2O).b) To determine the surface tension of glycerol/acetic acid/aniline solutions at differentConcentrations and construction of graph.2. Part II11 marksa) Preparation of Sodium acetate-acetic acid buffer solutions of different pHb) Preparation of Ammonium chloride-ammonium hydroxide buffer solutions of differentpHc) pH metric titration of strong acid/strong base,d) pH metric titration of weak acid/strong base.3. Viva – voce2 marks4. Regularity in maintenance of Lab Notebook2 marks5. Attendance5 marksReference Books: Khosla, B. D.; Garg, V. C. & Gulati, A. Senior Practical Physical Chemistry, R. Chand &Co.: New Delhi (2011). Halpern, A. M. & McBane, G. C. Experimental Physical Chemistry 3rd Ed.; W. H.Freeman & Co.: New York (2003). Nad, A.K., Mahapatra, B., Ghoshal, A., An Advanced Course in Practical Chemistry,New Central Book Agency (P) Ltd., Kolkata, India. Das, Subhas C., Advanced Practical Chemistry for 3-Year Honours Course.Assam University: Silchar.CBCS UG CHEMISTRYPage 14 of 99
CBCS: B. Sc. (Honours) with CHEMISTRYCORE COURSECHEMISTRY(Honours)(2nd Semester)Course No.:CHEMISTRY-C-201(Organic Chemistry – I)Hydrocarbons and StereochemistryContact Hours: 60; Credits: 04Full Marks 70[End Semester Exam (50) Internal Assessment (20)]Pass Marks 28 [End Semester Exam (20) Internal Assessment(08)](In the End-semester examination, two questions of 10 marks will be set from each unit,one needs to be answered from each unit)Objective of this course is to impart knowledge on basic organic chemistry, hydrocarbon,stereochemistry and conformational analysisUnit 1: Basics of Organic ChemistryOrganic Compounds: Classification, and Nomenclature, Hybridization, Shapes of molecules,Influence of hybridization on bond properties.Electronic Displacements: Inductive, electromeric, resonance and mesomeric effects,hyperconjugation and their applications; Dipole moment; Organic acids and bases; theirrelative strength. Homolytic and Heterolytic fission with suitable examples. Curly arrowrules, formal charges; Electrophiles and Nucleophiles; Nucleophlicity and basicity; Types,shape and their relative stability of Carbocations, Carbanions, Free radicals and Carbenes.Introduction to types of organic reactions and their mechanism: Addition, Elimination andSubstitution reactions.Unit 2: Chemistry of Aliphatic HydrocarbonsFormation of alkanes, Wurtz Reaction, Wurtz-Fittig Reactions, Free radical substitutions:Halogenation -relative reactivity and selectivity.Formation of alkenes and alkynes by elimination reactions, Mechanism of E1, E2, E1cbreactions. Saytzeff and Hofmann eliminations.Electrophilic additions their mechanisms (Markownikoff / Anti Markownikoff addition),ozonolysis, reduction (catalytic and chemical). 1, 2-and 1, 4-addition reactions in conjugateddienes and, Diels-Alder reaction; Allylic and benzylic bromination and mechanism, e.g.propene, 1-butene, toluene, ethyl Benzene.Acidity, Electrophilic and Nucleophilic additions. Hydration to form carbonyl compounds,Alkylation of terminal alkynes.Assam University: Silchar.CBCS UG CHEMISTRYPage 15 of 99
Unit 3: Aromatic and Polynuclear HydrocarbonsAromaticity: Hückel’s rule, aromatic character of arenes, cyclic carbocations/carbanions andheterocyclic compounds with suitable examples. Electrophilic aromatic substitution:halogenation, nitration, sulphonation and Friedel-Craft’s alkylation/acylation with theirmechanism. Directing effects of the groups.Reactions of naphthalene phenanthrene and anthracene Structure, Preparation and structureelucidation and important derivatives of naphthalene and anthracene; Polynuclearhydrocarbons.Unit 4: StereochemistryFischer Projection, Newmann and Sawhorse Projection formulae and their inter-conversions;Geometrical isomerism: cis–trans and, syn-anti isomerism E/Z notations with C.I.P rules.Optical Isomerism: Optical Activity, Specific Rotation, Chirality/Asymmetry, Enantiomers,molecules with two or more chiral-centres, diastereoisomers, meso structures, racemicmixture and resolution. Relative and absolute configuration: D/L and R/S designations.Unit 5: Cycloalkanes and Conformational AnalysisTypes of cycloalkanes and their relative stability, Baeyer strain theory, Conformationanalysis of alkanes: Relative stability: Energy diagrams of cyclohexane, monosubstituted,1,2-, 1,3-, 1,4-Disubstitutedcyclohexane: Chair, Boat and Twist boat forms; Relative stabilitywith energy diagrams, Strain-less ring theory.Reference Books: Morrison, R. N. & Boyd, R. N. Organic Chemistry, Dorling Kindersley (India) Pvt. Ltd.(Pearson Education). Finar, I. L. Organic Chemistry (Volume 1), Dorling Kindersley (India) Pvt. Ltd. (PearsonEducation). Finar, I. L. Organic Chemistry (Volume 2: Stereochemistry and the Chemistry of NaturalProducts), Dorling Kindersley (India) Pvt. Ltd. (Pearson Education). Eliel, E. L. & Wilen, S. H. Stereochemistry of Organic Compounds; Wiley: London, 1994. Kalsi, P. S. Stereochemistry Conformation and Mechanism; New Age International, 2005.Assam University: Silchar.CBCS UG CHEMISTRYPage 16 of 99
CBCS: B. Sc. (Honours) with CHEMISTRYCORE COURSECHEMISTRY LAB(Honours)(2nd Semester)Course No.:CHEMISTRY-C-201-LAB(Organic Chemistry – I)PracticalContact Hours: 60; Credits: 02Full Marks 30Pass Marks 12Examination Time: 12 hours (spread over two days)During examination, Two experiments (one from each of the Part I and Part II), to beassigned to each student by drawing lots.1. Part I: Purification:08 10 marks(a)Phthalic acid / Benzoic acid from hot water (using fluted filter paper and stemless funnel)(b)Acetanilide from boiling water(c)Naphthalene/ m-Dinitrobenzene from ethanol(d)Naphthalene/ camphor/phthalic acid (by sublimation)2. Part II: Chromatographic separation1611 marks(a)2,4-Dinitrophenyl hydrazones of any two carbonyl compounds (e.g.,benzophenone and benzyl; p-nitrobenzaldehyde and benzaldehyde) from theirmixture and determination of Rf values (By Thin layer chromatography)(b)Paper chromatographic separation and determination of Rf values of mixture ofany three amino acids from their mixture (alanine, glycine and leucine or anyother set). Spray reagent: Ninhydrin.3. Viva – voce2 marks4. Regularity in maintenance of Lab Notebook2 marks5. Attendance5 marksReference Books: Vogel, A. I., A Textbook of Qualitative Organic Analysis, ELBS. Nad, A.K., Mahapatra, B., Ghoshal, A., An Advanced Course in Practical Chemistry,New Central Book Agency (P) Ltd., Kolkata, India. Furniss, B. S.; Hannaford, A. J.; Smith, P. W. G.; Tatchell, A. R., Practical OrganicChemistry, 5 th Ed., Pearson (2012) Mann, F. G. & Saunders, B. C., Practical Organic Chemistry, Pearson Education (2009) Das, Subhas C., Advanced Practical Chemistry for 3-Year Honours Course.Assam University: Silchar.CBCS UG CHEMISTRYPage 17 of 99
CBCS: B. Sc. (Honours) with CHEMISTRYCORE COURSECHEMISTRY(Honours)(2nd Semester)Course No.:CHEMISTRY-C-202(Physical Chemistry – II)Chemical Thermodynamics and its ApplicationsContact Hours: 60; Credits: 04Full Marks 70[End Semester Exam (50) Internal Assessment (20)]Pass Marks 28 [End Semester Exam (20) Internal Assessment(08)](In the End-semester examination, two questions of 10 marks will be set from each unit,one needs to be answered from each unit)Objective of this course is to provide insight on Chemical Thermodynamics, theirmathematical expressions and ApplicationsUnit 1: Chemical Thermodynamics IIntensive and extensive variables; state and path functions; isolated, closed and open systems;zeroth law of thermodynamics.First law: Concept of heat (q), work (w), internal energy (U), and statement of first law;enthalpy (H), relation between heat capacities, calculations of q, w, U and H for reversible,irreversible and free expansion of gases (ideal and van der Waals) under isothermal andadiabatic conditions.Thermochemistry: Heats of reactions: standard states; enthalpy of formation of molecules andions and enthalpy of combustion and its applications; calculation of bond energy, bonddissociation energy and resonance energy from thermochemical data, effect of temperature(Kirchhoff’s equations) and pressure on enthalpy of reactions. Adiabatic flame temperature,explosion temperature.Unit 2: Chemical Thermodynamics IISecond Law: Concept of entropy; thermodynamic scale of temperature, statement of thesecond law of thermodynamics; molecular and statistical interpretation of entropy.Calculation of entropy change for reversible and irreversible processes.Third Law: Statement of third law, concept of residual entropy, calculation of absoluteentropy of molecules.Free Energy Functions: Gibbs and Helmholtz energy; variation of S, G, A with T, V, P; Freeenergy change and spontaneity. Relation between Joule-Thomson coefficient and otherthermodynamic parameters; inversion temperature; Gibbs-Helmholtz equation; Maxwellrelations; thermodynamic equation of state.Assam University: Silchar.CBCS UG CHEMISTRYPage 18 of 99
Unit 3: Systems of Variable CompositionPartial molar quantities, dependence of thermodynamic parameters on composition; GibbsDuhem equation, chemical potential of ideal mixtures, change in thermodynamic functions inmixing of ideal gases.Unit 4: Chemical EquilibriumCriteria of thermodynamic equilibrium, degree of advancement of reaction, chemicalequilibria in ideal gases, concept of fugacity. Thermodynamic derivation of relation betweenGibbs free energy of reaction and reaction quotient. Coupling of exoergic and endoergicreactions. Equilibrium constants and their quantitative dependence on temperature, pressureand concentration. Free energy of mixing and spontaneity; thermodynamic derivation ofrelations between the various equilibrium constants Kp, Kc and Kx. Le Chatelier’s Principle(quantitative treatment); equilibrium between ideal gases and a pure condensed phase.Unit 5: Solutions and Colligative PropertiesDilute solutions; lowering of vapour pressure, Raoult’s and Henry’s Laws and theirapplications. Excess thermodynamic functions.Thermodynamic derivation using chemical potential to derive relations between the fourcolligative properties [(i) relative lowering of vapour pressure, (ii) elevation of boiling point,(iii) Depression of freezing point, (iv) osmotic pressure] and amount of solute. Applicationsin calculating molar masses of normal, dissociated and associated solutes in solution.Reference Books: Peter, A. & Paula, J. de. Physical Chemistry 9th Ed., OUP (2011). Castellan, G. W. Physical Chemistry 4th Ed., Narosa (2004). Engel, T. & Reid, P. Physical Chemistry 3 rd Ed., Prentice-Hall (2012). McQuarrie, D. A. & Simon, J. D. Molecular Thermodynamics, Viva Books Pvt. Ltd.: NewDelhi (2004). Assael, M. J.; Goodwin, A. R. H.; Stamatoudis, M.; Wakeham, W. A. & Will, S.Commonly Asked Questions in Thermodynamics, CRC Press: NY (2011). Levine, I. N. Physical Chemistry 6 th Ed., Tata Mc Graw Hill (2010). Metz, C. R. 2000 Solved Problems in Chemistry, Schaum Series (2006)Assam University: Silchar.CBCS UG CHEMISTRYPage 19 of 99
CBCS: B. Sc. (Honours) with CHEMISTRYCORE COURSECHEMISTRY LAB(Honours)(2nd Semester)Course No.:CHEMISTRY-C-202-LAB(Physical Chemistry – II)PracticalContact Hours: 60; Credits: 02Full Marks 30Pass Marks 12Examination Time: 12 hours (spread over two days)During examination, two experiments to be assigned to each student by drawing lots1. Physical Experiments:10.
Modified as per CBCS Syllabus Structure Preparation Committee Meeting, March 28, 2017 . Contents 1. CBCS UG - Course Curriculum – Basic Structure 2. CBCS UG – SYLLABI for CHEMISTRY (HONOURS) . [Core for BSc with Chemistry] and GE [Hons with other Discipline] in accordance with sections 2.3, 2.1 and
UGC-NET Paper 1st Unit-1 www.ugc-net.com pg. 4 Logical memory: to learn something by using intellect and it’s recalling when needed. Important Points on Understanding level of teaching(ULT) Memory level teaching is the prerequisite for the understanding level of teaching as it talks about the generaliza
were under the UGC scales of pay as on 1.1.2016 and recruited thereafter and fulfilled all the eligibility criteria laid down by the UGC for the purpose in the UGC Revised Pay Scales, 2016. (5) The UGC Revised Pay Scales, 2016 are applicable to the teachers who were in the scales other than UGC
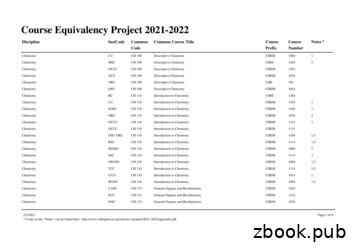
Chemistry ORU CH 210 Organic Chemistry I CHE 211 1,3 Chemistry OSU-OKC CH 210 Organic Chemistry I CHEM 2055 1,3,5 Chemistry OU CH 210 Organic Chemistry I CHEM 3064 1 Chemistry RCC CH 210 Organic Chemistry I CHEM 2115 1,3,5 Chemistry RSC CH 210 Organic Chemistry I CHEM 2103 1,3 Chemistry RSC CH 210 Organic Chemistry I CHEM 2112 1,3
UGC NET Paper 1st Unit-2 www.ugc-net.com pg. 1 UGC NTA NET Unit-2 Research Aptitude Research means to search the existing thing 'something' but remains unknown to us. So, Research means to find out something new on the topic that already existed. It is a searching process for finding new information about the things we already know.
Physical chemistry: Equilibria Physical chemistry: Reaction kinetics Inorganic chemistry: The Periodic Table: chemical periodicity Inorganic chemistry: Group 2 Inorganic chemistry: Group 17 Inorganic chemistry: An introduction to the chemistry of transition elements Inorganic chemistry: Nitrogen and sulfur Organic chemistry: Introductory topics
Accelerated Chemistry I and Accelerated Chemistry Lab I and Accelerated Chemistry II and Accelerated Chemistry Lab II (preferred sequence) CHEM 102 & CHEM 103 & CHEM 104 & CHEM 105 General Chemistry I and General Chemistry Lab I and General Chemistry II and General Chemistry Lab II (with advisor approval) Organic chemistry, select from: 9-10
CHEM 0350 Organic Chemistry 1 CHEM 0360 Organic Chemistry 1 CHEM 0500 Inorganic Chemistry 1 CHEM 1140 Physical Chemistry: Quantum Chemistry 1 1 . Chemistry at Brown equivalent or greater in scope and scale to work the studen
Apr 06, 2020 · University Grants Commission: The UGC was established in 1953 and made into a statutory organisation with the UGC Act in 1956. UGC is responsible for coordinating, determining and maintaining standards of higher education. The University Grants Com