Organic Chemistry 3rd Edition Klein Test Bank
Organic Chemistry 3rd Edition Klein Test BankFull Download: ry-3rd-edition-klein-test-bank/Klein, Organic Chemistry 3eChapter 21.What is the molecular formula for the following compound?A.B.C.D.E.C2H6OC4H6OC4H10OC2H4ONone of theseAnswer: CLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy2.Which of the following compounds have a molecular formula of C 2H6O?A.B.C.D.E.IIIIIIIVBoth I and IIIAnswer: ELearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy3.Which of the following is the correct condensed structure for the followingcompound?This sample only, Download all chapters at: alibabadownload.com
CH2CH2OCH3CH3CH3CHCH2OHAnswer: CLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy4.Which of the following is the correct condensed structure for the HClCH3C2H4CH3OHClAnswer: BLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy5.Which of the following is the correct condensed structure for the followingcompound?
A.B.C.D.E.CH2 CH(CH2)3C(CH3)3CH(CH2)4C(CH3)3(CH3)2CH(CH2) 4CH3CH2CH(CH2)3C(CH3)3(CH)3(CH2)3C(CH3)3Answer: ALearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Medium6.Which of the following is the correct condensed structure for the C(CH2)3C(CH3)2CH3(CH3)3C2(CH2)3CH3CH3C C(CH2)3C(CH3)3CH3CC(CH2)3C(CH3)3Answer: DLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Medium
7.Which of the following is the correct condensed structure for the BrCH(CH3)2Answer: ELearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Medium8.Provide the correct condensed structure for the following compound.Answer: (CH3)3C(CH2)2OCH(CH2CH3)2Learning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Hard9.Provide the correct condensed structure for the following compound.
Answer: (CH3)2N(CH2)3CH(CH3)2Learning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Hard10.Which of the following is the correct molecular formula for r: CLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy11.Which of the following is the correct Lewis structure for CH3(CH2)2NH2?
A.B.C.D.E.IIIIIIIVBoth II and IIIAnswer: CLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy12.Which of the following is the correct Lewis structure for CH3(CH2)2OH?A.B.C.D.E.IIIIIIIVBoth II and IIIAnswer: BLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy13.Which of the following is the correct Lewis structure for (CH3)2CHCH2OH?
A.B.C.D.E.IIIIIIIVBoth III and IVAnswer: CLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy14.Which of the following is the correct Lewis structure for (CH3)3C(CH2)2NHCH3?
A.B.C.D.E.IIIIIIIVVAnswer: DLearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Medium15.Draw the Lewis structure for CH3C C(CH2)3C(CH3)3.Answer:Learning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Medium
16.Draw the Lewis structure for (CH3)3C(CH2)2OCH(CH2CH3)2.Answer:Learning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Hard17.Identify the partially condensed structure for CH3CH2OCH2CH3.A.B.C.D.E.IIIIIIIVVAnswer: ALearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy
18.Identify the partially condensed structure for CH3CH2CH2NH2.Answer: ALearning Objective: 2.1 Convert molecular representations from one drawing style toanother, including Lewis structures, partially condensed structures, condensedstructures, and molecular formulasDifficulty: Easy19.Which of the following bond-line structures are of the same compound?A.B.C.D.E.I and IIII and IIIIII and IVII and IVNone of theseAnswer: DLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy20.Which of the following bond-line structures are of the same compound?
A.B.C.D.E.I and IIIII and IIIIII and IVII and IVNone of theseAnswer: ALearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy21.How many hydrogen atoms are connected to the indicated carbon atom?A.B.C.D.E.onetwothreefournoneAnswer: ELearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy22.How many hydrogen atoms are connected to the indicated carbon atom?A.B.onetwo
C.D.E.threefournoneAnswer: ALearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy23.How many hydrogen atoms are connected to the indicated carbon atom?A.B.C.D.E.onetwothreefournoneAnswer: BLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy24.How many hydrogen atoms are connected to the indicated carbon atom?A.B.C.D.E.onetwothreefournoneAnswer: A
Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy25.How many hydrogen atoms are connected to the indicated carbon atom?A.B.C.D.E.onetwothreefournoneAnswer: ELearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy26.For the following equation, how many hydrogen atoms are added or lost?A.B.C.D.E.added oneadded twolost onelost twono changeAnswer: DLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy
27.For the following equation, how many hydrogen atoms are added or lost?A.B.C.D.E.added oneadded twolost onelost twono changeAnswer: BLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy28.For the following equation, how many hydrogen atoms are added or lost?A.B.C.D.E.added oneadded twolost onelost twono changeAnswer: ELearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy29.For the following equation, how many hydrogen atoms are added or lost?
A.B.C.D.E.added oneadded twolost onelost twono changeAnswer: ELearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy30.For the following equation, how many hydrogen atoms are added or lost?A.B.C.D.E.added oneadded twolost onelost twono changeAnswer: DLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Medium31.For the following equation, how many hydrogen atoms are added or lost?
A.B.C.D.E.added oneadded twolost onelost twono changeAnswer: ELearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Medium32.Which of the following is the correct Lewis structure for the following compound?A.B.C.D.E.IIIIIIIVnone of theseAnswer: BLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy
33.Which of the following is the correct bond-line structure for (CH3)4C?A.B.C.D.E.IIIIIIIVNone of theseAnswer: CLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy34.Which of the following is the correct bond-line structure for (CH3)2CHCH2CH3?A.B.C.D.E.IIIIIIIVNone of theseAnswer: ALearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Easy
35.Which of the following(CH3)2CHCH2C(CH3)3?A.B.C.D.E.IIIIIIIVNone of theseisthecorrectbond-linestructureforAnswer: BLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Medium36.Which of the followingCH3C C(CH2)2CH(CH3)2?A.B.C.D.E.IIIIIIIVNone of theseisthecorrectbond-linestructureforAnswer: DLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Medium
37.Which of the following isCH3CHOH(CH2)2CH(CH2CH3) orAnswer: BLearning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Medium38.Draw a bond-line structure for CH3CH2O(CH2)2CH(CH3)2.Answer:Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard
39.Draw a bond-line structure for (CH3)2N(CH2)3CH(CH3)2.Answer:Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard40.Draw a bond-line structure for CH3C C(CH2)3C(CH3)2CH2OCH3.Answer:Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard41.Draw a bond-line structure for each constitutional isomer with a molecularformula of C2H4O.Answer:Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard42.Draw a bond-line structure for each constitutional isomer with a molecularformula of C3H8O.
Answer:Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard43.Provide a condensed structure for the following compound.Answer: (CH3)3CCH2CH(CH2CH3)(CH2)2CH(CH3)CH2CH3Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard44.Provide a condensed structure for the following compound.Answer: (CH3)3C(CH2)2CH(NH2)CH CHCH3Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard
45.Draw a bond-line structure for each constitutional isomer with molecular formulaC4H10O.Answer:Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard46.Draw a bond-line structure for each constitutional isomer with molecular formulaC4H11N.Answer:Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Medium47.Naproxen, sold under the trade name Aleve, has the following structure. What isthe molecular formula for naproxen?
Answer: C14H14O3Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard48.AZT, used in the treatment of AIDS, has the following structure. What is themolecular formula for AZT?Answer: C10H13N5O4Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard49.Capsaicin, found in peppers, has the following structure. What is the molecularformula for capsaicin?
Answer: C18H27NO3Learning Objective: 2.2 Demonstrate how to read and draw bond-line structuresthrough converting other styles of molecular representation into bond-line structures andvice versaDifficulty: Hard50.Which of the following compounds contain an alcohol functional group?A.B.C.D.E.IIIIIIIVNone of theseAnswer: CLearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy51.Which of the following compounds contain an alkene functional group?
A.B.C.D.E.IIIIIIIVNone of theseAnswer: ALearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy52.Which of the following compounds contain an amine functional group?A.B.C.D.E.IIIIIIIVNone of theseAnswer: DLearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy53.Which of the following compounds contain a ketone functional group?
A.B.C.D.E.IIIIIIIVAll of theseAnswer: ALearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy54.Which of the following compounds contain an aromatic ring?A.B.C.D.E.IIIIIIIVBoth III and IVAnswer: CLearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy55.Which of the following compounds contain an ester functional group?
A.B.C.D.E.IIIIIIIVBoth I and IVAnswer: ALearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy56.Which of the following compounds contain an amide functional group?A.B.C.D.E.IIIIIIIVBoth II and IIIAnswer: BLearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy57.Which of the following compounds contain an anhydride functional group?A.B.C.D.E.IIIIIIIVnone of the aboveAnswer: CLearning Objective: 2.3 Identify and draw the functional groups
Difficulty: Easy58.Which of the following compounds contain an alkyne functional group?A.B.C.D.E.IIIIIIIVnone of the aboveAnswer: ALearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy59.Which of the following compounds contain a thiol functional group?A.B.C.D.E.IIIIIIIVnone of the aboveAnswer: BLearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy60.Which of the following compounds contain an alkyl halide functional group?
A.B.C.D.E.IIIIIIIVnone of the aboveAnswer: DLearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Easy61.What functional group(s) is (are) present in the following compound?A.B.C.D.E.ketone and alkeneketone and alkynealdehyde and alkenealdehyde and alkyneester and alkeneAnswer: CLearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Medium62.Which of the following compounds have both a ketone and an ester functionalgroup?
A.B.C.D.E.IIIIIIIVVAnswer: ELearning Objective: 2.3 Identify and draw the functional groupsDifficulty: Medium63.Norethynodrel, a component of the first combined oral contraceptive, has thefollowing structure. Identify the functional groups in Norethynodrel.Answer:
Learning Objective: 2.3 Identify and draw the functional groupsDifficulty: Medium64.Identify the functional groups in the following compound.Answer:Learning Objective: 2.3 Identify and draw the functional groups
Difficulty: Medium65.Tamiflu , the most effective antiviral drug used to treat avian influenza, has thefollowing structure. Identify the functional groups in Tamiflu .Answer:Learning Objective: 2.3 Identify and draw the functional groupsDifficulty: Hard66.Aspartame, an artificial sweetener used in Equal and diet beverages, has thefollowing structure. Identify the functional groups in Aspartame.
Answer:Learning Objective: 2.3 Identify and draw the functional groupsDifficulty: Hard67.Draw all the constitutional isomers with a molecular formula of C3H6O and labelthe functional groups in each isomer.Answer:
Learning Objective: 2.3 Identify and draw the functional groupsDifficulty: Hard68.Amoxicillin, an antibiotic, has the following structure. Identify the functionalgroups in amoxicillin.Answer:Learning Objective: 2.3 Identify and draw the functional groupsDifficulty: Hard69.Viracept, used in the treatment of HIV, has the following structure. Identify thefunctional groups in Viracept.
Answer:Learning Objective: 2.3 Identify and draw the functional groupsDifficulty: Hard70.How many hydrogen atoms are connected to the indicated carbon atom?
A.B.C.D.E.onetwothreefournoneAnswer: BLearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy71.How many hydrogen atoms are connected to the indicated carbon atom?A.B.C.D.E.onetwothreefournoneAnswer: ELearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy72.How many hydrogen atoms are connected to the indicated carbon atom?A.B.C.D.E.onetwothreefournoneAnswer: ALearning Objective: 2.4 Identify formal charges on carbon
Difficulty: Easy73.How many hydrogen atoms are connected to the indicated carbon atom?A.B.C.D.E.onetwothreefournoneAnswer: BLearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy74.How many hydrogen atoms are connected to the indicated carbon atom?A.B.C.D.E.onetwothreefournoneAnswer: ELearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy75.How many hydrogen atoms are connected to the indicated carbon atom?
A.B.C.D.E.onetwothreefournoneAnswer: ALearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy76.What is the formal charge on the indicated carbon atom?A.B.C.D.E.-2-10 1 2Answer: DLearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy77.What is the formal charge on the indicated carbon atom?A.B.C.D.E.-2-10 1 2Answer: DLearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy
78.What is the formal charge on the indicated carbon atom?A.B.C.D.E.-2-10 1 2Answer: DLearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy79.What is the formal charge on the indicated carbon atom?A.B.C.D.E.-2-10 1 2Answer: BLearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy80.What is the formal charge on the indicated carbon atom?A.-2
B.C.D.E.-10 1 2Answer: BLearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy81.What is the formal charge on the indicated carbon atom?A.B.C.D.E.-2-10 1 2Answer: BLearning Objective: 2.4 Identify formal charges on carbonDifficulty: Easy82.What is the formal charge on the oxygen atom in the following compound?A.B.C.D.E. 1 2-1-20Answer: ALearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy
83.What is the formal charge on the nitrogen atom in the following compound?A.B.C.D.E.-1-2 1 20Answer: CLearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy84.What is the formal charge on the nitrogen atom in the following compound?A.B.C.D.E. 1 2-1-20Answer: ALearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy85.What is the formal charge on the indicated oxygen atom in the followingcompound?
A.B.C.D.E. 1 2-1-20Answer: ELearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy86.What is the formal charge on the nitrogen atom in the following compound?A.B.C.D.E. 1 2-1-20Answer: ELearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy87.Which of the following compounds have 1 as a formal charge on an oxygenatom?A.B.III
C.D.E.IIIIVBoth I and IVAnswer: BLearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Medium88.Which of the following compounds have 1 as a formal charge on the nitrogenatom?A.B.C.D.E.IIIIIIIVBoth I and IIAnswer: DLearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Medium89.Determine the formal charges on each atom except hydrogen.Answer:
Learning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Medium90.Diazomethane has the molecular formula CH2N2. Draw the preferred Lewisstructure for diazomethane and assign formal charges to all atoms excepthydrogen.Answer:Learning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Hard91.Draw Lewis structure for the following compound.Answer:
Learning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Medium92.How many lone pairs of electrons are on the oxygen atom?A.B.C.D.E.onetwothreefournoneAnswer: CLearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy93.How many lone pairs of electrons are on the nitrogen atom?A.B.C.D.E.onetwothreefournoneAnswer: B
Learning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy94.How many lone pairs of electrons are on the oxygen atom?A.B.C.D.E.onetwothreefournoneAnswer: BLearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy95.How many lone pairs of electrons are on the nitrogen atom?A.B.C.D.E.onetwothreefournoneAnswer: ALearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy
96.How many lone pairs of electrons are on the nitrogen atom?A.B.C.D.E.onetwothreefournoneAnswer: ELearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy97.How many lone pairs of electrons are on the indicated oxygen atom?A.B.C.D.E.onetwothreefournoneAnswer: BLearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy98.How many lone pairs of electrons are on the indicated oxygen atom?
A.B.C.D.E.onetwothreefournoneAnswer: ALearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Medium99.How many total lone pairs of electrons are in the following compound?A.B.C.D.E.onetwothreefournoneAnswer: CLearning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Easy100.Draw all lone pairs of electrons for the following compound.
Answer:Learning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Medium101.Draw all lone pairs of electrons for the following compound.Answer:
Learning Objective: 2.5 Describe the relationship between the number of bonds, thenumber of lone pairs, and formal charge for oxygen and nitrogen atomsDifficulty: Medium102.The indicated bond in the following compound is of the paper.A.B.C.D.in the planeout of the planebehind the planeNone of theseAnswer: BLearning Objective: 2.6 Describe how wedges and dashes are used to indicate threedimensional molecular structureDifficulty: Easy103.The indicated bond in the following compound is of the paper.A.B.C.D.in the planeout of the planebehind the planeNone of theseAnswer: CLearning Objective: 2.6 Describe how wedges and dashes are used to indicate threedimensional molecular structureDifficulty: Easy104.The indicated bond in the following compound is of the paper.
A.B.C.D.in the planeout of the planebehind the planeNone of theseAnswer: CLearning Objective: 2.6 Describe how wedges and dashes are used to indicate threedimensional molecular structureDifficulty: Easy105.Which of the following is a Fischer projection?A.B.C.D.E.IIIIIIIVBoth III and IVAnswer: BLearning Objective: 2.6 Describe how wedges and dashes are used to indicate threedimensional molecular structureDifficulty: Easy
106.Which of the following is a Haworth projection?A.B.C.D.E.IIIIIIIVBoth III and IVAnswer: CLearning Objective: 2.6 Describe how wedges and dashes are used to indicate threedimensional molecular structureDifficulty: Easy107.Label the bonds that are out of the plane and behind the plane of the paper.Answer:
Learning Objective: 2.6 Describe how wedges and dashes are used to indicate threedimensional molecular structureDifficulty: Medium108.Which of the following pairs are resonance structures of each other?A.B.C.D.E.IIIIIIIVNone of theseAnswer: DLearning Objective: 2.7 Define "resonance," describing the relationship betweenresonance and molecular orbital theoryDifficulty: Easy109.Which of the following pairs are resonance structures of each other?A.B.C.D.E.IIIIIIIVNone of theseAnswer: C
Learning Objective: 2.7 Define "resonance," describing the relationship betweenresonance and molecular orbital theoryDifficulty: Easy110.The spreading of positive or negative charge over two or more atoms in acompound is called ocalizationNone of theseAnswer: BLearning Objective: 2.7 Define "resonance," describing the relationship betweenresonance and molecular orbital theoryDifficulty: Easy111.Delocalization of charge over two or more atoms a sstabilizesNone of theseAnswer: DLearning Objective: 2.7 Define "resonance," describin
Klein, Organic Chemistry 3e Chapter 2 1. What is the molecular formula for the following compound? A. C 2 H 6 O B. C 4 H 6 O C. C 4 H 10 O D. C 2 H 4 O E. None of these Answer: C Learning Objective: 2.1 Co
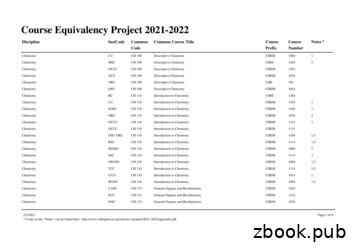
Chemistry ORU CH 210 Organic Chemistry I CHE 211 1,3 Chemistry OSU-OKC CH 210 Organic Chemistry I CHEM 2055 1,3,5 Chemistry OU CH 210 Organic Chemistry I CHEM 3064 1 Chemistry RCC CH 210 Organic Chemistry I CHEM 2115 1,3,5 Chemistry RSC CH 210 Organic Chemistry I CHEM 2103 1,3 Chemistry RSC CH 210 Organic Chemistry I CHEM 2112 1,3
Textbook Essentials of Organic Chemistry by Dewick The following textbooks are also available in the chemistry library on reserve: Organic Chemistry: A Short Course by Hart, Craine, Hart and Hadid Introduction to Organic Chemistry by Brown and Poon Fundamentals of Organic Chemistry by McMurry Essential Organic Chemistry by Bru
In Press Advanced Inorganic Chemistry Gurdeep Raj 06 270 Chromatography B.K. Sharma 06 Organic Chemistry 302 Advanced Organic Chemistry Aditi Singhal 08 279 Organic Chemistry Natural Products - Vol. I O. P. Agarwal 09 280 Organic Chemistry Natural Products - Vol. II O. P. Agarwal 09 281 Organic Chemistry Reactions & Reagents O. P. Agarwal 10
ORGANIC CHEMISTRY 10/e Francis A. Carey Robert M. Giuliano McGraw Hill 2017. TEXTBOOK ORGANIC CHEMISTRY 9/e Francis A. Carey Robert M. Giuliano McGraw Hill 2013. TEXTBOOK ORGANIC CHEMISTRY EIGHTH EDITION F. A. Carey and R. M. Giuliano McGraw-Hill New York 2011. TEXTBOOK ORGANIC CHEMISTRY
David Klein. 2. Organic Chemistry as a Second Language David Klein (any edition is fine). Recommended for students that struggles in General Chemistry or have had a long break from taking chemistry
CHEM 0350 Organic Chemistry 1 CHEM 0360 Organic Chemistry 1 CHEM 0500 Inorganic Chemistry 1 CHEM 1140 Physical Chemistry: Quantum Chemistry 1 1 . Chemistry at Brown equivalent or greater in scope and scale to work the studen
Klein’s presence at a seminar in economics given by Neyman in Griffith Evans’ home (Reid 1982, p.168). Klein was accepted for the recently opened Ph.D. program in economics at MIT from 1942 and got Paul Samuelson as supervisor. 2 Klein chose Keynesian theory as topic for a PhD thesis. Klein had
Physical chemistry: Equilibria Physical chemistry: Reaction kinetics Inorganic chemistry: The Periodic Table: chemical periodicity Inorganic chemistry: Group 2 Inorganic chemistry: Group 17 Inorganic chemistry: An introduction to the chemistry of transition elements Inorganic chemistry: Nitrogen and sulfur Organic chemistry: Introductory topics