Food Chemistry - Chris Beat Cancer
Food Chemistry 112 (2009) 374–380Contents lists available at ScienceDirectFood Chemistryjournal homepage: www.elsevier.com/locate/foodchemAntiproliferative and antioxidant activities of common vegetables: Acomparative studyDominique Boivin a, Sylvie Lamy a, Simon Lord-Dufour a, Jessica Jackson a, Edith Beaulieu a, Martine Côté c,Albert Moghrabi b, Stéphane Barrette b, Denis Gingras a, Richard Béliveau a,b,*,1aLaboratoire de Médecine Moléculaire, Centre de Cancérologie Charles-Bruneau, Hôpital Ste-Justine, Université du Québec à Montréal, 3175 Côte Ste-Catherine, Montréal,Québec, Canada H3T 1C5bService d’hématologie-oncologie, Centre de Cancérologie Charles-Bruneau, Hôpital Ste-Justine, 3175 Chemin Côte-Ste-Catherine, Montréal, Québec, Canada H3T 1C5cMinistère de l’Agriculture, des Pêcheries et de l’Alimentation du Québec, 200, chemin Sainte-Foy, Québec, Canada G1R 4X6a r t i c l ei n f oArticle history:Received 12 February 2008Received in revised form 5 May 2008Accepted 23 May 2008Keywords:Cancer preventionCruciferous vegetablesAllium vegetablesAntioxidantsa b s t r a c tEpidemiological studies have consistently linked abundant consumption of fruits and vegetables to areduction of the risk of developing several types of cancer. In most cases, however, the identificationof specific fruits and vegetables that are responsible for these effects is still lacking, retarding the implementation of effective dietary-based chemopreventive approaches. As a first step towards the identification of foods endowed with the most potent chemopreventive activities, we evaluated the inhibitoryeffects of extracts isolated from 34 vegetables on the proliferation of 8 different tumour cell lines. Theextracts from cruciferous vegetables as well as those from vegetables of the genus Allium inhibited theproliferation of all tested cancer cell lines whereas extracts from vegetables most commonly consumedin Western countries were much less effective. The antiproliferative effect of vegetables was specific tocells of cancerous origin and was found to be largely independent of their antioxidant properties. Theseresults thus indicate that vegetables have very different inhibitory activities towards cancer cells and thatthe inclusion of cruciferous and Allium vegetables in the diet is essential for effective dietary-based chemopreventive strategies.! 2008 Elsevier Ltd. All rights reserved.1. IntroductionIt is currently estimated that dietary factors account for approximately one third of cancer death, similar to the impact of smoking(Doll & Peto, 1981). Such a close relationship between diet and cancer is well illustrated by the large variations in rates of specific cancers among countries and by the observations that these rates arestrongly correlated with differences in several aspects of the diet(Doll & Peto, 1981; Willett, 2002). Among the dietary factors thatare most closely linked to cancer, a large number of populationbased studies have consistently shown that individuals who eatfive servings or more of fruits and vegetables daily have approximately half the risk of developing a wide variety of cancer types,particularly those of the gastrointestinal tract (Gescher, Pastorino,Plummer, & Manson, 1998; World Cancer Research Fund & AmerAbbreviations: ORAC, oxygen radical absorbance capacity* Corresponding author. Address: Laboratoire de Médecine Moléculaire, Centrede Cancérologie Charles-Bruneau, Hôpital Ste-Justine, 3175 Côte Ste-Catherine,Montréal, Québec, Canada H3T 1C5. Tel.: 1 514 345 2366; fax: 1 514 345 2359.E-mail address: molmed@recherche-ste-justine.qc.ca (R. Béliveau).1Holder of the ‘‘Chaire en prévention et traitement du cancer” from UQÀM and ofthe ‘‘Chaire Claude-Bertrand en Neurochirurgie” from Université de Montréal.0308-8146/ - see front matter ! 2008 Elsevier Ltd. All rights reserved.doi:10.1016/j.foodchem.2008.05.084ican Institute for Cancer Research, 1997). These chemopreventiveproperties of fruits and vegetables arise from their high contentof phytochemicals such as phenolic compounds (Naczk & Shahidi,2004, 2006) that target several key events involved in the development of cancer (Dorai & Aggarwal, 2004; Surh, 2003). Potentialmechanisms for cancer prevention of phytochemicals include prevention of DNA adduct formation (Ames, Gold, & Willett, 1995), enhanced carcinogen elimination (Talalay, 2000), inhibition ofinflammatory processes (Surh et al., 2001), interference with tumour angiogenesis (Lamy, Gingras, & Béliveau, 2002; Tosetti, Ferrari, De Flora, & Albini, 2002), as well as through a directcytotoxic effect on tumour cells (Martin, 2006). This pleiotropicmechanism of action of phytochemicals imply that the chemopreventive properties that are associated with fruits and vegetablesconsumption are complex and likely arise from synergistic combinations from several distinct molecules, not only within a givenfood but also from the overall composition of the diet (Lee, Lee,& Lee, 2004; Liu, 2003; McCullough & Giovannucci, 2004). Clearly,the identification of specific foods or food groups that have beneficial effects on certain types of cancer represent an important issuein order to bonify current chemopreventive strategies based on increased consumption of fruits and vegetables.
D. Boivin et al. / Food Chemistry 112 (2009) 374–380As a first step towards this goal, we have undertaken a systematic evaluation of the chemopreventive potential of several vegetables by monitoring their antiproliferative effects on a wide varietyof tumour cell lines. Surprisingly, we found that there are considerable differences in the ability of vegetables to inhibit the proliferation of various cancer cells. We observed that this anticancerproperty is preferentially associated with cruciferous and Alliumvegetables, whereas most vegetables commonly consumed in Western countries have a much weaker antiproliferative effects. Theseresults illustrate the need to improve current dietary recommendations by actively promoting increased consumption of cruciferous and Allium vegetables as an essential means to reduce theincidence of cancer.2. Materials and methods2.1. ChemicalsFluorescein sodium salt, 2,20 -azobis (2-methylpropionamidine)dihydrochloride (AAPH) and 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox) were purchased from Sigma–Aldrich (Oakville, ON, Canada).2.2. Preparation of vegetable juicesFresh vegetables were obtained from local producers (Montreal,QC, Canada) in May–July 2005, stored at 4 "C, and processed within24 h. Juices were prepared by passing 100 g of vegetables througha domestic centrifugal juice extractor (Juiceman Professional series210, model JM210C, Montreal, QC, Canada). The liquid obtainedwas clarified by centrifugation at 50,000g, 45 min at 4 "C. Thesupernatant was then sterilized by filtration through a 0.22-lm filter, and aliquots were immediately frozen in liquid nitrogen. Protein concentrations were determined by the Bradford methodusing the Coomassie Plus assay kit (Pierce).2.3. Cell cultureAll cell lines were cultured at 37 "C under a humidified atmosphere containing 5% CO2. AGS (stomach adenocarcinoma, ATCCCRL-1739) were cultured in F12-K medium containing 10% FBS;MCF-7 (mammary gland adenocarcinoma, ATCC HTB-22) were cultured in MEM containing 0.01 mg/ml insulin and 10% FBS; Panc-1(pancreatic carcinoma ATCC CRL-1469) were cultured in DMEMhigh glucose containing 10% FBS; PC-3 (prostatic adenocarcinoma,ATCC CRL-1435) were cultured in Ham’s F12 containing 10% calfserum; A549 (lung carcinoma, ATCC CCL-185) cells were culturedin DMEM low glucose containing 10% calf serum; Daoy (medulloblastoma, ATCC HTB-186) and U-87 MG (glioblastoma, ATCCHTB-14) cells were cultured in MEM containing 10% FBS; Caki-2(renal carcinoma, ATCC HTB-47) cells were cultured in McCoy’s5A medium containing 10% FBS. NHDF (normal human dermalfibroblasts) were cultured in FGM-2 medium (Clonetics) containing 1 ng/ml hFGF, 5 lg/ml insulin, and 2% FBS.2.4. Cell proliferation assayCells were plated in 96-well plates at 2500–5000 cells/well in200 ll complete medium and incubated at 37 "C under a humidified atmosphere containing 5% CO2 for 24 h. The next day, the medium was removed and replaced by 100 ll fresh medium containing1% FCS and the specified concentrations of juices. Cell viability wasdetermined by assaying the mitochondrial activity of treated cellsafter a 48 h incubation, with the highly sensitive WST-1 assay.Briefly, 10 ll of tetrazolium salt WST-1 reagent was added to each375well and the soluble formazan dye produced by metabolically active cells was monitored every minute for 30 min at 37 "C on aSpectraMax Plus reader (molecular devices).2.5. Oxygen radical absorbance capacity (ORAC) assayThe ORAC-fluorescein assay was performed essentially as described previously (Dávalos, Gómez-Cordovés, & Bartolomé,2004) with minor modifications. Briefly, 20 ll of antioxidant (vegetable extracts or Trolox standards), and 120 ll of 0.117 lM fluorescein in 75 mM phosphate buffer, pH 7.4 were pipetted intothe well of the microplate. The mixture was preincubated for15 min at 37 "C, and then 60 ll of 40 mM AAPH were added rapidlyusing an electronic multichannel pipette. The fluorescence(kex 485 nm; kem 520 nm) was recorded every min for 80 minusing a SpectraMAXTM Gemini fluorescence plate reader (MolecularDevices). Calibration solutions of Trolox (0.5–8 lM) were also carried out in each assay. Data were exported from the SoftMax Pro3.1 software to Excel (Microsoft) for further calculations. The areaunder the fluorescence decay curve (AUC) was calculated asAUC 1 f1/f0 f2/f0 !!! f80/f0 where f0 is the initial fluorescenceat t 0 and fi the fluorescence at t i. ORAC-FL values were expressed as Trolox equivalents by using a standard curve andregression analysis performed using the Prism 4.0 software(GraphPad Software, San Diego, CA).3. Results3.1. Inhibition of tumour cell proliferation by vegetable extractsAs a first step towards the identification of vegetables containing antiproliferative activities towards cancer cells, the inhibitoryeffects of extracts from a wide variety of commonly consumed vegetables on eight different tumour cell lines derived from stomach,kidney, prostate, breast, brain, pancreatic and lung cancer wereexamined. There was considerable differences in the sensitivityof these cell lines to the vegetable extracts (Fig. 1). Tumour cellsderived from prostate and stomach cancer were most sensitive tothe extracts while cells from kidney, pancreatic and lung cancerswere much less affected by the tested extracts. For example, 23of the 34 the tested vegetable extracts inhibited the proliferationof prostate tumour cells by more than 50%, while only 7 extractswere active against kidney cancer cells (Table 1).In addition to cruciferous vegetables, all members of the Alliumfamily tested in this study were powerful inhibitors of tumour cellproliferation. In fact, among all vegetables tested in this study, theextract from garlic was by far the strongest inhibitor of tumour cellproliferation, with complete growth inhibition of all tested celllines. Leek, immature (green) and mature (yellow) onions werealso highly inhibitory against most cell lines, although green onionwas less active against tumour cells from the kidney, while yellowonion was a modest inhibitor of the lung tumour cells and had nosignificant inhibitory activity against kidney tumour cells. Overall,these results indicate that there is substantial differences in theantiproliferative properties of vegetables against tumour cellsand that cruciferous, dark green and Allium vegetables are endowed with potent anticancer properties (Table 2).The potency of inhibition of cell proliferation by cruciferous andAllium vegetables was then investigated, using serial dilutions ofthe extracts (Fig. 2a). Garlic was the most potent inhibitor of cellproliferation with a marked reduction of U-87 glioblastoma cellproliferation at a 1/1000 (corresponding to 3.32 mg raw vegetable/ml) dilution of the extract. Brussels sprouts extracts alsostrongly inhibited the proliferation of these cells, with 30% inhibition at a 1/1000 dilution (3.32 mg raw vegetable/ml) and complete
376D. Boivin et al. / Food Chemistry 112 (2009) 374–380Fig. 1. Inhibition of tumour cell proliferation by vegetable extracts. Tumour cell lines derived from stomach adenocarcinoma (AGS), mammary gland adenocarcinoma (MCF7), pancreatic carcinoma (Panc-1), prostatic adenocarcinoma (PC-3), lung carcinoma (A549), medulloblastoma (Daoy), renal carcinoma (Caki-2), and glioblastoma (U-87 MG)were incubated for 48 h in the absence or in the presence of a 1/20 dilution (corresponding to 166 mg raw vegetable per ml) of the indicated vegetable extracts. Cell viabilitywas determined by assaying the mitochondrial activity of treated cells using the WST-1 assay. Results are the means SD of 4 experiments performed in triplicates.
D. Boivin et al. / Food Chemistry 112 (2009) 374–380377Table 1Inhibition of cancer cell proliferation by vegetable juicesCancer cell lineNumber of vegetable extracts ( 50% inhibition)PC-3 (prostate)AGS (stomach)U-87 (glioblastoma)DAOY (medulloblastoma)MCF-7 (breast)A-549 (lung)Panc-1 (pancreas)Caki-2 le 2Identification of vegetables with chemopreventive potentialLittleIntermediateInhibitory effect on cancer cell proliferationaAcorn squashCeleryBock choyEggplantBoston lettuceCarrotEndiveEnglish cucumberFennel bulbJalapenoOrange sweet pepperPotatoRadicchioRomaine lettuceTomatoHighVery Green beanRadishRed cabbageRutabagaYellow onionBrussel sproutsCabbageCurly cabbageGarlicGreen onionKaleLeekSpinachaLittle activity is defined as an inhibitory effect of 50% on less than 2 cell lines.Intermediate activity is defined as an inhibitory effect of 50% on more than 2 butless than 4 cell lines. High activity is defined as an inhibitory effect of 50% on fourand more cell lines. Very high activity is defined as an inhibitory effect of at least50% on all cell lines.inhibition at 1/100 dilution of the extract (33.2 mg raw vegetable/ml). The inhibitory effect of Brussels sprouts was slightly higherthan that of green onion (60% at 1/100 dilution), while matureonion and broccoli inhibited proliferation by 30% at a 1/100 dilution, with complete inhibition being only observed at a 1/20 dilution (166 mg raw vegetable/ml).In order to determine whether the strong inhibitory effects ofcruciferous and Allium vegetables were specific for tumour cells,the effects of these extracts on the proliferation of normal fibroblast were subsequently monitored. As shown in Fig. 2b, all extracts had strong inhibitory activities against a glioblastoma cellline but had negligible effects on the growth of normal cells,strongly suggesting that the antiproliferative properties of thesevegetables are specific to cells of tumour origin.3.2. Antiproliferative and antioxidant activitiesOxidative stress is now recognized as a major factor associatedwith the development of chronic diseases, including cancer andcardiovascular disease (Ames et al., 1995). This has led to thehypothesis that the beneficial effects of fruits and vegetables couldbe largely explained by their high content of antioxidants (Prior,2003). Antioxidant activity is involved in cancer prevention atthe initiation stage while antiproliferative activity is targeting cancer cells at the promotion and progression stages (Manson, 2003;Surh, 2003). We measured the oxygen radical absorbance capacity(ORAC) of the extracts (Table 3), and found that there were significant differences between ORAC values of various vegetables. Garlic, curly cabbage and Brussels sprouts were the strongest source ofantioxidants (41.1, 40.5, and 32.9 lmol Trolox equiv./ml, respectively) whereas other vegetables such as lettuce and cucumbercontained considerably less antioxidants (1.5 and 1.4 lmol TroloxFig. 2. Specificity of the antiproliferative activities of vegetable extracts againsttumour cells. (a) Glioblastoma cells were incubated for 48 h with 1/20, 1/100, 1/500, 1/1000 and 1/5000 dilutions (corresponding to 166, 33.2, 6.64, 3.32 and0.66 mg raw vegetable per ml, respectively) of the indicated vegetable extracts andthe extent of proliferation was monitored as described in the legend to Fig. 1. (b)Glioblastma cells (dark columns) or NHDF normal fibroblasts (open columns) wereincubated with the indicated extracts and proliferation was determined. Arepresentative experiment is shown.equiv./ml). Our results suggest that both antioxidant and antiproliferative activities, involved in two different mechanisms of chemoprevention, could be considered for a better evaluation of theglobal anticancer potential of fruits and vegetables. The resultsare in agreement with reports showing that many powerful anticancer vegetables, including cruciferous vegetables such as broccoli and cauliflower, show modest antioxidant activities in vitro(Wu et al., 2004).4. DiscussionOver 250 epidemiological studies have suggested that individuals consuming diets high in fruits and vegetables have a reducedrisk of developing several cancers (World Cancer Research Fund &American Institute for Cancer Research, 1997). These observationsform the basis of current recommendations from governmentalhealth agencies around the world promoting consumption of atleast 5 servings of these foods daily as a mean to reduce the inci-
378D. Boivin et al. / Food Chemistry 112 (2009) 374–380Table 3Antioxidant capacity, antiproliferative activity and protein concentration of vegetablejuicesVegetableextractsAntioxidant capacity(lmol Trolox equiv./mL)Antiproliferativeactivity(% inhibition)Proteinconcentration(mg/mL)GarlicCurly cabbageBrusselssproutsBeetrootRed cabbageFiddleheadSpinachEggplantYellow onionGreen onionKaleAsparagusOrange gaCauliflowerCabbageJalapenoGreen beansRomainelettuceFennel bulbEndiveBock choyCarrotCeleryTomatoBoston lettuceAcorn squashEnglishCucumber41.1 1.940.5 2.732.9 0.510095 51009.15 0.331.75 0.074.94 0.2324.7 1.823.2 0.419.4 0.717.4 0.515.5 2.011.5 0.110.6 0.59.7 1.29.2 1.27.9 0.310094 264 6991 699 210010071 10 11.54 0.051.10 0.020.76 0.015.45 0.270.95 0.040.45 0.031.02 0.014.32 0.102.16 0.020.24 0.016.9 0.26.8 0.96.4 0.15.9 0.15.8 0.045.1 0.34.8 0.44.7 0.34.7 0.23.9 0.22,9 0.225 410041 84 910071 110010049 488 30 17.08 0.223.71 0.020.92 0.050.75 0.013.44 0.080.75 0.021.66 0.151.27 0.051.13 0.033.70 0.031.71 0.052.7 0.42.5 0.12.1 0.51.9 0.41.8 0.21.6 0.11.5 0.31.5 0.31.4 0.22 100 119 11 670 216 10 1253 1041 110.79 0.010.44 0.011.02 0.031.30 0.020.44 0.020.07 0.010.99 0.030.74 0.020.27 0.02dence of chronic diseases and cancer (Heimendinger & Chapelsky,1996). However, fruits and vegetables contain varying levels ofchemopreventive phytochemicals so that a global protective roleof these foods is unlikely (McCullough & Giovannucci, 2004)and that increased consumption of certain foods with the highestphytochemical content must also be strongly encouraged (Johnston, Taylor, & Hampl, 2000). In this respect, in a large prospective study, total fruit and vegetable consumption was notassociated with a reduction of the overall cancer incidence (Hunget al., 2004). Interestingly, a strong reduction of bladder cancerassociated with the consumption of cruciferous vegetables wasobserved within this same male cohort (Michaud et al., 1999),again suggesting that specific foods or foods groups have benefitsagainst specific cancers. This is supported by numerous studiesshowing that consumption of cruciferous vegetables (Talalay &Fahey, 2001; Verhoeven, Goldbohm, van Poppel, Verhagen, &van den Brandt, 1996), Allium vegetables (Fleischauer
D. Boivin et al./Food Chemistry 112 (2009) 374–380 375. Fig. 1. Inhibition of tumour cell proliferation by vegetable extracts. Tumour cell lines derived from stomach adenocarcinoma (AGS), mammary gland adenocarcinoma (MCF-a
So Beat It! Just Beat It, Beat It! f f E5 D5 E5 D5 E5 D5 Voice Voice Vln. 1 Vc. Fl. Alto Sax. Ten. Sax. E. Gtr. E. Bass E. Bass Pno. Synth. Dr. kick you, then they beat you, then they'll tell you it's fair, so Beat It! But you wan-na be bad. Just Beat It, Beat It! No one wants to be de feat 63 D Beat It!
Put the beat in your arms, put the beat in your arms, Put the beat in your arms. Way up high, way down low, Put the beat in your arms. 5. Put the beat in your head, put the beat in your head, Put the beat in your head. Nodding “Yes” and shaking “No” Put the beat in your head. *Descend
Dotted Quarter Beat Unit; More Rhythms 15.1. Building Aural/Oral Skills Subdivided and Borrowed Beat in Compound Meter Divide the class into two groups. The first group taps the beat and chants the division of beat (eighths); the second group taps the beat and chants the subdivided beat. Exchange tasks.
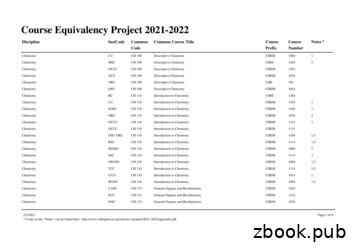
Chemistry ORU CH 210 Organic Chemistry I CHE 211 1,3 Chemistry OSU-OKC CH 210 Organic Chemistry I CHEM 2055 1,3,5 Chemistry OU CH 210 Organic Chemistry I CHEM 3064 1 Chemistry RCC CH 210 Organic Chemistry I CHEM 2115 1,3,5 Chemistry RSC CH 210 Organic Chemistry I CHEM 2103 1,3 Chemistry RSC CH 210 Organic Chemistry I CHEM 2112 1,3
56 Ben's Dream Chris Van Allsburg 57 Jumanji Chris Van Allsburg 58 Just a Dream Chris Van Allsburg 59 Probuditi! Chris Van Allsburg 60 Queen of the Falls Chris Van Allsburg 61 The Garden of Abdul Gasazi Chris Van Allsburg 62 The Misadventures of Sweetie Pie Chris Van Allsburg 63 The Mysteries of Harris Burdick Chris V
Music as a Second Language & The Modern Band Movement – Little Kids Rock Teacher Manual v3.3 DRUMS drum chart Library BEAT 18 BEAT 24 BEAT 10 BEAT 11 BEAT 12 BEAT 37 q q q q xxxxxxxx ÊÊ q E q q xxxxxxxx Ê q E q q xxxxxxxx ÊÊ q q q q q xxxxxxxx ÊÊ EÊ q xxxxxxxx q q q q q q q q xxxxxxxx Iconic Notation Standard Notation
Ovarian cancer is the seventh most common cancer among women. There are three types of ovarian cancer: epithelial ovarian cancer, germ cell cancer, and stromal cell cancer. Equally rare, stromal cell cancer starts in the cells that produce female hormones and hold the ovarian tissues together. Familial breast-ovarian cancer
Physical chemistry: Equilibria Physical chemistry: Reaction kinetics Inorganic chemistry: The Periodic Table: chemical periodicity Inorganic chemistry: Group 2 Inorganic chemistry: Group 17 Inorganic chemistry: An introduction to the chemistry of transition elements Inorganic chemistry: Nitrogen and sulfur Organic chemistry: Introductory topics