Chapter 10 Coordination Chemistry II: Bonding
Chapter 10 Coordination Chemistry II:Bonding10-1 Experimental Evidence for Electronic Structures10-2 Theories of Electronic Structure10-3 Ligand Field Theory10-4 Angular Overlap10-5 The Jahn-Teller Effect10-6 Four- and Six-Coordinate Preferences10-7 Other Shapes“Inorganic Chemistry” Third Ed. Gary L. Miessler, Donald A. Tarr, 2004, Pearson Prentice Hallhttp://en.wikipedia.org/wiki/Expedia
Experimental Evidence for Electronic StructuresThermodynamic DataMagnetic SusceptibilityElectronic SpectraCoordination Numbers and MolecularShapes
Experimental Evidence for Electronic Structures;Thermodynamic DataOne of the primary goal of a bonding theory is toexplain the energy of compound.The energy is openly not determined directly byexperiment.Thermodynamic measurements of enthalpies andfree energies of reaction are used to compare.Bonding strength Stability constants(formation constants)
Experimental Evidence for Electronic Structures;Thermodynamic DataWhat is the stability constants?The equilibrium constants for formation ofcoordination complex.
Experimental Evidence for Electronic Structures;Thermodynamic DataStability constants HSAB conceptsThermodynamic values Prediction of properties, structures
Experimental Evidence for Electronic Structures;Thermodynamic DataHSAB conceptsThe gist of this theory is that soft acids react faster and formstronger bonds with soft bases, whereas hard acids reactfaster and form stronger bonds with hard bases, all other factorsbeing equal.The classification in the original work was mostly based onequilibrium constants for reaction of two Lewis bases competingfor a Lewis acid.Hard acids and hard bases tend to have:small sizehigh oxidation statelow polarizabilityhigh electronegativityenergy low-lying HOMO (bases) or energy high-lying LUMO(acids).
Experimental Evidence for Electronic Structures;Thermodynamic DataHSAB concepts
Experimental Evidence for Electronic Structures;Thermodynamic DataChelating LigandsEntropy Effecten vs methyl amineChelate EffectFive or six membered ringFigure in head .Stability .
Experimental Evidence for Electronic Structures;Magnetic SusceptibilityThe magnetic properties of a coordinationcompound can provide indirect evidence of theorbital energy level.Hund’s rule the max. # of unpaired e-.Diamagnetic: all e- paried repelled by a magnetic fieldParamagnetic: all e- paried attracted into a magnetic fieldMagnetic Susceptibility: Measuring Magnetism
Experimental Evidence for Electronic Structures;Magnetic SusceptibilityMagnetic SusceptibilityGouy methodA sample that is to be tested is suspendedfrom a balance between the poles of amagnet. The balance measures theapparent change in the mass of thesample as it is repelled or attracted by themagnetic field.
Experimental Evidence for Electronic Structures;Magnetic SusceptibilityIn physics and applied disciplines such as electricalengineering, the magnetic susceptibility is the degree ofmagnetization of a material in response to an appliedmagnetic field.Electron spin Spin magnetic moment (ms)Total spin magnetic moment Spin quantum # S (sum of ms)Isolated oxygen atom 1s22s2p4S 1/2 1/2 1/2 -1/2 1Electron spin Orbital magnetic moment (ml)Total orbital magnetic moment Orbital quantum # L (sum of ml)Max. L for the p4L 1 0 -1 1 1
Experimental Evidence for Electronic Structures;Magnetic SusceptibilityTwo sources of magnetic moment – spin (S) and Angular (L) motions of electronsSpin quantum numberAngular momentum quantum numberThe equation for the magnetic momentContribution from L is small in first transition series2.00023 2
Experimental Evidence for Electronic Structures;Electronic SpectraGive a direct evidence of orbital energy levelGive an information for geometry of complexes
Theories of Electronic StructureValence bond theoryCrystal field theoryLigand field theoryAngular overlap method
Theories of Electronic Structure;Valence bond theoryHybridization ideasOctahedral: d2sp3d orbitals could be 3d or 4d for the first-rowtransition metals. (hyperligated, hypoligated)
Theories of Electronic Structure;Valence bond theoryFe(III)Isolated ion; 5 unpaired eIn Oh compound; 1 or 5 unpaired eCo(II)Low spinLow spinHigh spinHigh spin
Theories of Electronic Structure;Crystal field theoryCrystal field theory (CFT) is a model that describes theelectronic structure of transition metal compounds, all ofwhich can be considered coordination complexes.CFT successfully accounts for some magnetic properties,colours, hydration enthalpies, and spinel structures oftransition metal complexes, but it does not attempt todescribe bonding.CFT was developed by physicists Hans Bethe and JohnHasbrouck van Vleck in the 1930s.CFT was subsequently combined with molecular orbitaltheory to form the more realistic and complex ligand fieldtheory (LFT), which delivers insight into the process ofchemical bonding in transition metal complexes.
Theories of Electronic Structure;Crystal field theoryRepulsion between d-orbital electrons and ligand electrons Splitting of energy levels of d-orbitals
Theories of Electronic Structure;Crystal field theory
Theories of Electronic Structure;Crystal field theory
Theories of Electronic Structure;Crystal field theoryElectrostatic approachIn an Octahedral field of ligand e- pairs; any ein them are repelled by the field.Crystal field stabilization energy (CFSE);the actual distribution vs the uniform field.Good for the concept of the repulsion oforbitals by the ligands but no explanationfor bonding in coordination complexes.
Theories of Electronic Structure;Crystal field theory
Theories of Electronic Structure;Crystal field theory
Theories of Electronic Structure;Crystal field theory
Theories of Electronic Structure;Crystal field theory
Theories of Electronic Structure;Crystal field theory
Theories of Electronic Structure;Crystal field theory
Theories of Electronic Structure;Crystal field theoryWhy are complexes formed in crystal field theory?Crystal Field Stabilization Energy (CFSE)Or Ligand Field Stabilization Energy (LFSE) the stabilization of the d orbitals because ofmetal-ligand environments
Theories of Electronic Structure;Crystal field theory E strong field – weak field E 0 weak field E 0 strong field
Theories of Electronic Structure;Crystal field theoryWhat determine ?Depends on the relative energiesof the metal ions and ligandorbitals and on the degree ofoverlap.
Theories of Electronic Structure;Crystal field theorySpectrochemical Series for Metal IonsOxidation # Only low spin aqua complexSmall size & higher chargePt4 Ir3 Pd4 Ru3 Rh3 Mo3 Mn4 Co3 Fe3 V2 Fe2 Co2 Ni2 Mn2
Ligand field theory;Molecular orbitals for Octahedral complexesCFT & MO were combinedThe dx2-y2 and dz2 orbitals can form bonding orbitalswith the ligand orbitals, but dxy, dxz, and dyz orbitalscannot form bonding orbitals
Ligand field theory;Molecular orbitals for Octahedral complexesThe combination ofthe ligand and metalorbitals (4s, 4px, 4py,4pz, 3dz2, and 3dx2-y2)form six bonding andsix antibonding witha1g, eg, t1u symmetries.The metal T2g orbitalsdo Electronnot havein bondingorbitals providetheappropriatesymmetrypotential energy that holds- nonbondingmolecules together
Ligand field theory;Orbital Splitting and Electron SpinStrong-field ligand – Ligands whose orbitalsinteract strongly with the metal orbitals large oWeak-field ligand.d0 d3 and d8 d10 – only one electronconfiguration possible no difference in thenet spinStrong fields lead to low-spin complexesWeak fields lead to high-spin complexes
Ligand field theory;Orbital Splitting and Electron SpinWhat determine ?Depends on the relativeenergies of the metal ionsand ligand orbitals and onthe degree of overlap.
Ligand field theory;Orbital Splitting and Electron SpinSpectrochemical Series for Metal IonsOxidation # Small size & higher chargePt4 Ir3 Pd4 Ru3 Rh3 Mo3 Mn4 Co3 Fe3 V2 Fe2 Co2 Ni2 Mn2
Ligand field theory;Ligand field Stabilization Energy
Ligand field theory;Orbital Splitting and Electron SpinOrbital configuration of the complex isdetermined by o, πc, and πeIn general o for 3 ions is larger than o for 2 ionswith the same # of e-. o π low-spin o π high-spinFor low-spinconfigurationRequire a strongfield ligand
Ligand field theory;Ligand field Stabilization Energy
Ligand field theory;Orbital Splitting and Electron SpinThe position of the metal in the periodictableSecond and third transition series form lowspin more easily than metals form the firsttransition series-The greater overlap between the larger 4dand 5d orbitals and the ligand orbitals-A decreased pairing energy due to thelarger volume available for electrons
Ligand field theory;Pi-BondingThe reducible representation is
Ligand field theory;Pi-BondingLUMO orbitals:can be usedfor π bonding with metalHOMO
Ligand field theory;Pi-Bondingmetal-to-ligand π bondingor π back-bonding-Increase stability-Low-spin configuration-Result of transfer ofnegative charge away fromthe metal ionLigand-to metal π bonding-decrease stability-high-spin configuration
Ligand field theory;Square planar Complexes; Sigma bonding
Ligand field theory;Square planar Complexes; Sigma bondingll e- from metal16 e-8 e-
Ligand field theory;Tetrahedral Complexes; Sigma bondingThe reducible representation isA1 and T2
Ligand field theory;Tetrahedral Complexes; Pi bondingThe reducible representation isE, T1 and T2
Angular OverlapLFT No explicit use of the energy change that resultsDifficult to use other than octahedral, squareplanar, tetrahedral.Deal with a variety of possible geometries andwith a mixture of ligand. Angular OverlapModelThe strength of interaction between individual ligandorbitals and metal d orbitals based on the overlapbetween them.
Angular Overlap:Sigma-Donor InteractionsThe strongest σ interactionThere are no examples of complexes with e- inthe antibonding orbitals from s and p orbitals,and these high-energy antibonding orbitals arenot significant in describing the spectra ofcomplexes. we will not consider them further.
Angular Overlap:Sigma-Donor Interactions
Angular Overlap:Sigma-Donor InteractionsStabilization is 12eσ
Angular Overlap:Pi-Acceptor InteractionsThe strongest π interaction is considered tobe between a metal dxy orbitals and a ligand π*orbital.Because of the overlap for these orbitals issmaller than the σ overlap, eπ eσ.
Angular Overlap:Pi-Acceptor Interactions
Angular Overlap:Pi-Acceptor Interactions
Angular Overlap:Pi-Donor InteractionsIn general, in situations involving ligands that canbehave as both π acceptors and π donors (suchas CO, CN-), the π acceptor nature predominates.
Angular Overlap:Pi-Donor Interactions
Angular Overlap:Pi-Acceptor Interactions
Angular Overlap:Types of the ligands and the spectrochemical seriesSpectrochemical Series for LigandsCO CN- PPh3 NO2- phen bipy en σ donor onlyNH3 py CH3CN NCS- H2O C2O42OH- RCO2- F- N3- NO3- Cl- SCNS2- Br- Iπ acceptor (strong field ligand)π donor(weak field ligand)
Angular Overlap:Magnitudes of eσ eπ and Metal and ligand
Angular Overlap:Magnitudes of eσ eπ and Angular overlapparameters derivedfrom electronicspectraeσ is always largerthan eπ. overlapisoelectronicThe magnitudes ofboth the σ and πparameters with size and electronegativity ofthe halide ions.overlap
Angular Overlap:Magnitudes of eσ eπ and Can describe theelectronic energyof complexes withdifferent shapes orwith combinationsof different liagnds.The magnitude of o Magneticproperties andvisible spectrum.
Angular Overlap:The Jahn-Teller EffectThere cannot be unequal occupation of orbitals with identical orbitals.To avoid such unequal occupation, the molecule distorts so thatthese orbitals no longer degenerate.In other words, if the ground electron configuration of a nonlinearcomplex is orbitally degenerate, the complex will distort to removethe degeneracy and achieve a lower energy.
Angular Overlap:The Jahn-Teller Effect
Angular Overlap:Four- and Six-Coordinate PreferenceAngular overlap calculationsOnly σ bonding is considered.Low-spin square planarLarge # of bonds formed inthe octahedral complexes.
Angular Overlap:Four- and Six-Coordinate Preference
Angular Overlap:Four- and Six-Coordinate PreferenceHow accurate are these predictions?Their success is variable, because of there are other differencesbetween metals and between ligands.In addition, bond lengths for the same ligand-metal pair depend onthe geometry of the complex.The interactions of the s and p orbitals.The formation enthalpy for complexes also becomes more negativewith increasing atomic number and increasing ionization energy.By careful selection of ligands, many of the transition metal ions canform compounds with geometries other than octahedral.
Angular Overlap:Other shapes111Strength of σ–interaction112 3/49/89/800
Angular Overlap:Other shapesTrigonal-bipyramidal ML5 (D3h) σ-donor only
Angular Overlap LFT No explicit use of the energy change that results Difficult to use other than octahedral, square planar, tetrahedral. Deal with a variety of possible geometries and with a mixture of ligand. Angular Overlap Model The strength of interaction between individual ligand orbitals and metal d orbitals based on the overlap
Part One: Heir of Ash Chapter 1 Chapter 2 Chapter 3 Chapter 4 Chapter 5 Chapter 6 Chapter 7 Chapter 8 Chapter 9 Chapter 10 Chapter 11 Chapter 12 Chapter 13 Chapter 14 Chapter 15 Chapter 16 Chapter 17 Chapter 18 Chapter 19 Chapter 20 Chapter 21 Chapter 22 Chapter 23 Chapter 24 Chapter 25 Chapter 26 Chapter 27 Chapter 28 Chapter 29 Chapter 30 .
TO KILL A MOCKINGBIRD. Contents Dedication Epigraph Part One Chapter 1 Chapter 2 Chapter 3 Chapter 4 Chapter 5 Chapter 6 Chapter 7 Chapter 8 Chapter 9 Chapter 10 Chapter 11 Part Two Chapter 12 Chapter 13 Chapter 14 Chapter 15 Chapter 16 Chapter 17 Chapter 18. Chapter 19 Chapter 20 Chapter 21 Chapter 22 Chapter 23 Chapter 24 Chapter 25 Chapter 26
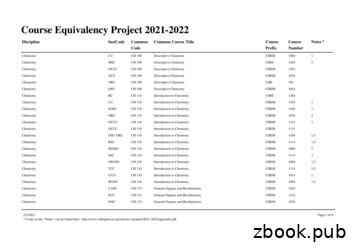
Chemistry ORU CH 210 Organic Chemistry I CHE 211 1,3 Chemistry OSU-OKC CH 210 Organic Chemistry I CHEM 2055 1,3,5 Chemistry OU CH 210 Organic Chemistry I CHEM 3064 1 Chemistry RCC CH 210 Organic Chemistry I CHEM 2115 1,3,5 Chemistry RSC CH 210 Organic Chemistry I CHEM 2103 1,3 Chemistry RSC CH 210 Organic Chemistry I CHEM 2112 1,3
Coordination Chemistry!Coordination compound!Compound that contains 1 or more complexes!Example "[Co(NH 3) 6]Cl 3 "[Cu(NH 3) 4][PtCl 4] "[Pt(NH 3) 2Cl 2] Coordination Chemistry!Coordination sphere!Metal and ligands bound to it!Coordination number!number of donor atoms bonded to the central
Coordination Chemistry Coordination compounds neutral coordination compounds include the metal and its bound ligands the number of bonds in a coordination complex does not correlate with the formal oxidation state of the metal coordination complexes can also be charged –complex cations or complex anions in a complex ion,
DEDICATION PART ONE Chapter 1 Chapter 2 Chapter 3 Chapter 4 Chapter 5 Chapter 6 Chapter 7 Chapter 8 Chapter 9 Chapter 10 Chapter 11 PART TWO Chapter 12 Chapter 13 Chapter 14 Chapter 15 Chapter 16 Chapter 17 Chapter 18 Chapter 19 Chapter 20 Chapter 21 Chapter 22 Chapter 23 .
Physical chemistry: Equilibria Physical chemistry: Reaction kinetics Inorganic chemistry: The Periodic Table: chemical periodicity Inorganic chemistry: Group 2 Inorganic chemistry: Group 17 Inorganic chemistry: An introduction to the chemistry of transition elements Inorganic chemistry: Nitrogen and sulfur Organic chemistry: Introductory topics
Coordination Chemistry II: Isomers and Coordination Geometries Chapter 9 Monday, November 16, 2015. A Real World Example of Stuff from Class! Isomerism Coordination complexes often have a variety of isomeric forms. Structural I