Introductory ChemIstry - Pearson Education
IntroductoryChemistryTro IC5 FM i-xxxv v4.0.1.indd 112/11/13 1:04 PM
Tro IC5 FM i-xxxv v4.0.1.indd 212/11/13 1:04 PM
IntroductoryCHEMISTRyFifth EditionNivaldo J. TroWestmont CollegeBoston Columbus Indianapolis New York San Francisco Upper Saddle RiverAmsterdam Cape Town Dubai London Madrid Milan Munich Paris Montréal TorontoDelhi Mexico City São Paulo Sydney Hong Kong Seoul Singapore Taipei TokyoTro IC5 FM i-xxxv v4.0.1.indd 312/11/13 1:04 PM
Editor in Chief: Adam JaworskiAcquisitions Editor: Chris Hess, Ph.DDirector of Development: Jennifer HartExecutive Marketing Manager: Jonathan CottrellAssociate Team Lead, Program Management, Chemistry and Geosciences: Jessica MoroProgram Manager: Coleen MorrisonDevelopment Editor: Erin MulliganEditorial Assistant: Caitlin FalcoMarketing Assistant: Nicola HoustonTeam Lead, Project Management, Chemistry and Geosciences:Gina M. CheselkaProject Manager: Beth SweetenProduction Management: codeMantra, LLCCompositor: codeMantra, LLCIllustrator: Precision GraphicsImage Lead: Maya MelenchukPhoto Researcher: Peter Jardim, PreMedial GlobalText Permissions Manager: Michael FarmerText Permission Researcher: Electronic Publishing Services Inc.Interior Design Manager: Mark OngInterior Designer: Wanda Espana, WeeDesignCover Design Manager: Anthony GemmellaroCover Designer: Wanda Espana, WeeDesignOperations Specialist: Christy HallCover Art: Quade PaulCredits and acknowledgments borrowed from other sources and reproduced, with permission, in this textbookappear on the appropriate page within the text or on p. PC-1.Copyright 2015, 2011, 2009, 2006, 2003 Pearson Education, Inc., publishing as [imprint name and address]. Allrights reserved. Manufactured in the United States of America. This publication is protected by Copyright andpermission should be obtained from the publisher prior to any prohibited reproduction, storage in a retrievalsystem, or transmission in any form or by any means, electronic, mechanical, photocopying, recording, orlikewise. To obtain permission(s) to use material from this work, please submit a written request to PearsonEducation, Inc., Permissions Department, 1900 E. Lake Ave., Glenview, IL 60025. For information regardingpermissions, call (847) 486-2635.Many of the designations used by manufacturers and sellers to distinguish their products are claimed as trademarks. Where those designations appear in this book, and the publisher was aware of a trademark claim, thedesignations have been printed in initial caps or all caps.Library of Congress Cataloging-in-Publication DataTro, Nivaldo J.Introductory chemistry / Nivaldo J. Tro. —5th ed. volumes cmIncludes index.ISBN 978-0-321-91029-5 (extended) — ISBN 978-0-321-91873-4 (essentials)1. Chemistry—Textbooks. I. Title.QD33.2.T76 2015540—dc2320130352351 2 3 4 5 6 7 8 9 10—CRK—17 16 15 14www.pearsonhighered.comTro IC5 FM i-xxxv v4.0.1.indd 4ISBN 10: 0-321-91029-X;ISBN 13: 978-0-321-91029-512/11/13 1:04 PM
To AnnieAbout the AuthorNivaldo Tro,is a Professor of Chemistry at WestmontCollege in Santa Barbara, California, where he has been a faculty member since 1990. He received his Ph.D. in chemistry fromStanford University for work on developing and using opticaltechniques to study the adsorption and desorption of moleculesto and from surfaces in ultra high vacuum. He then went on tothe University of California at Berkeley, where he did post doctoral research on ultrafast reaction dynamics in solution. Sincecoming to Westmont, Professor Tro has been awarded grants fromthe American Chemical Society Petroleum Research Fund, fromResearch Corporation, and from the National Science Foundationto study the dynamics of various processes occurring in thin adlayer films adsorbed on dielectric surfaces. He has been honoredas Westmont's outstanding teacher of the year three times andhas also received the college's outstanding researcher of the yearaward. Professor Tro lives in Santa Barbara with his wife, Ann, and their four children, Michael, Ali, Kyle, and Kaden.In his leisure time, Professor Tro enjoys mountain biking, surfing, reading to his children, and being outdoors with hisfamily. vTro IC5 FM i-xxxv v4.0.1.indd 512/11/13 1:04 PM
Brief ContentsPreface12345678910111213141516171819The Chemical Worldxviii2Measurement and Problem Solving12Matter and Energy56Atoms and Elements94Molecules and Compounds128Chemical Composition166Chemical Reactions204Quantities in Chemical Reactions248Electrons in Atoms and the Periodic Table284Chemical Bonding324Gases358Liquids, Solids, and Intermolecular Forces410Solutions446Acids and Bases486Chemical Equilibrium528Oxidation and Reduction574Radioactivity and Nuclear Chemistry610Organic Chemistry642Biochemistry696Appendix: Mathematics ReviewMR-1GlossaryG-1Answers to Odd-Numbered ExercisesA-1Photo CreditsIndexPC-1I-1 viTro IC5 FM i-xxxv v4.0.1.indd 612/11/13 1:04 PM
ContentsPrefacexviii1The ChemicalWorld21.11.21.31.43556Soda Pop FizzChemicals Compose Ordinary ThingsAll Things Are Made of Atoms and MoleculesThe Scientific Method: How Chemists ThinkEveryday Chemistry Combustion and theScientific Method1.5A Beginning Chemist: How to SucceedChapter in reviewKey TermsExercises2Measurement andProblem Solving2.12.22.3Measuring Global TemperaturesScientific Notation: Writing Large and Small NumbersSignificant Figures: Writing Numbers to ReflectPrecision889101012131316Counting Significant Figures 17Exact Numbers 18Chemistry in the Media The COBE Satelliteand Very Precise Measurements That Illuminate OurCosmic Past192.42.52.6Significant Figures in Calculations20Multiplication and Division 20Rounding 20Addition and Subtraction 21Calculations Involving Both Multiplication/Divisionand Addition/Subtraction 22The Basic Units of Measurement24The Base Units 24Prefix Multipliers 25Derived Units 26Problem Solving and Unit ConversionConverting Between Units 27General Problem-Solving Strategy 29 Problem-Solving Procedure Solving UnitConversion Problems2.72.82.9Solving Multistep Unit Conversion ProblemsUnits Raised to a PowerChemistry and Health Drug DosageDensity 3031333436Calculating Density 36Density as a Conversion Factor 37Chemistry and Health Density,Cholesterol, and Heart Disease392.10 Numerical Problem-Solving Strategiesand the Solution Map Problem-Solving Procedure SolvingNumerical Problems27Chapter in reviewKey TermsExercises39 40414747 viiTro IC5 FM i-xxxv v4.0.1.indd 712/11/13 1:04 PM
viii Contents3.12 Energy and Heat Capacity CalculationsChapter in reviewKey TermsExercises4Atoms andElements4.14.24.34.44.54.64.74.83Matter andEnergy3.13.23.33.43.53.6In Your RoomWhat Is Matter?Classifying Matter According to Its State:Solid, Liquid, and GasClassifying Matter According to ItsComposition: Elements, Compounds,and MixturesDifferences in Matter: Physical andChemical PropertiesChanges in Matter: Physical andChemical Changes4.956575759606365Conservation of Mass: There Is No New Matter 67Energy68Chemistry in the Environment Getting Energy out of Nothing?69Key TermsExercises5Molecules andCompoundsMolecules and Atoms3.11 Temperature Changes: Heat CapacityEveryday Chemistry Coolers, Camping,and the Heat Capacity of WaterTro IC5 FM i-xxxv v4.0.1.indd 87799100101104108Sugar and SaltCompounds Display Constant CompositionChemical Formulas: How to 4A Molecular View of Elements and Compounds 1355.5Writing Formulas for Ionic Compounds71727695969797Polyatomic Ions in Chemical Formulas 133Types of Chemical Formulas 134Units of Energy 693.9 Energy and Chemical and Physical Change3.10 Temperature: Random Motion ofIsotopes: When the Number ofNeutrons VariesAtomic Mass: The Average Mass ofan Element’s AtomsChemistry in the Environment Chapter in review5.15.25.394Ions and the Periodic Table 109Radioactive Isotopes at Hanford, WashingtonSeparating Mixtures Through Physical Changes 673.73.8Experiencing Atoms at TiburonIndivisible: The Atomic TheoryEveryday Chemistry Atoms and HumansThe Nuclear AtomThe Properties of Protons, Neutrons,and ElectronsEveryday Chemistry Solid Matter?Elements: Defined by Their Numbersof ProtonsLooking for Patterns: The Periodic Lawand the Periodic TableIons: Losing and Gaining Electrons77818686Atomic Elements 135Molecular Elements 135Molecular Compounds 135Ionic Compounds 136138Writing Formulas for Ionic Compounds ContainingOnly Monoatomic Ions 138 Problem-Solving Procedure Writing Formulasfor Ionic Compounds 13812/11/13 1:04 PM
Contents ixWriting Formulas for Ionic Compounds Containing139Polyatomic Ions 5.65.75.85.9Nomenclature: Naming CompoundsNaming Ionic Compounds140140Naming Binary Ionic Compounds Containing aMetal That Forms Only One Type of Cation 141Naming Binary Ionic Compounds Containing a MetalThat Forms More Than One Type of Cation 142Naming Ionic Compounds Containing aPolyatomic Ion 143Everyday Chemistry Polyatomic Ions144Naming Molecular CompoundsNaming Acids145146Naming Binary Acids 146Naming Oxyacids 147Chemistry in the Environment Acid Rain5.10 Nomenclature Summary148148Ionic Compounds 149Molecular Compounds 149Acids 1495.11 Formula Mass: The Mass of a Moleculeor Formula UnitChapter in reviewKey .16.26.31671681696.46.5How Much Sodium?Counting Nails by the PoundCounting Atoms by the GramConverting between Moles andNumber of Atoms 169Converting between Grams and Molesof an Element 170Converting between Grams of an Element andNumber of Atoms 173Counting Molecules by the GramChemical Formulas as Conversion FactorsMass Percent Composition of CompoundsMass Percent Composition from aChemical FormulaTro IC5 FM i-xxxv v4.0.1.indd 96.86.9Calculating an Empirical Formula fromExperimental Data 186 Problem-Solving Procedure Obtaining anEmpirical Formula from Experimental Data 187Calculating Molecular Formulas forCompoundsKey TermsExercises188190196196174177Converting between Grams of a Compound and Molesof a Constituent Element 178Converting between Grams of a Compound andGrams of a Constituent Element 179Chlorine in Chlorofluorocarbons185Calculating Empirical Formulas for Compounds 185Chapter in reviewConverting between Grams and Molesof a Compound 174Converting between Grams of a Compound andNumber of Molecules 176Chemistry in the Environment 6.66.7Chemistry and Health Fluoridation ofDrinking de School Volcanoes, Automobiles, andLaundry DetergentsEvidence of a Chemical ReactionThe Chemical EquationHow to Write Balanced Chemical Equations205206209211 Problem-Solving Procedure Writing BalancedChemical Equations21212/11/13 1:04 PM
x Contents7.5Aqueous Solutions and Solubility:Compounds Dissolved in Water214Solubility 2157.67.77.87.9Precipitation Reactions: Reactions in AqueousSolution That Form a Solid218Predicting Precipitation Reactions 218 Problem-Solving Procedure Writing Equationsfor Precipitation Reactions220Writing Chemical Equations for Reactionsin Solution: Molecular, Complete Ionic, andNet Ionic EquationsAcid–Base and Gas Evolution Reactions221223Acid–Base (Neutralization) Reactions 223Gas Evolution Reactions 224Chemistry and Health NeutralizingExcess Stomach AcidOxidation–Reduction Reactions226226Combustion Reactions 2277.10 Classifying Chemical Reactions228Classifying Chemical Reactions byWhat Atoms Do 229Classification Flowchart 231Chemistry in the Environment The Reactions Involved in Ozone DepletionChapter in reviewKey TermsExercises8Quantities inChemical te Change: Too Much Carbon Dioxide249Making Pancakes: Relationships betweenIngredients250Making Molecules: Mole-to-Mole Conversions 251Making Molecules: Mass-to-Mass Conversions 253Chemistry in the Media The Controversyover Oxygenated Fuels254More Pancakes: Limiting Reactant,Theoretical Yield, and Percent Yield257Limiting Reactant, Theoretical Yield, andPercent Yield from Initial Masses of Reactants 260Enthalpy: A Measure of the Heat Evolvedor Absorbed in a Reaction264Everyday Chemistry Bunsen Burners2659Electrons in Atoms andthe Periodic Table2849.19.29.3285286288Treatment for Cancer9.49.5Key TermsExercisesTro IC5 FM i-xxxv v4.0.1.indd 10268272273The Bohr Model: Atoms with OrbitsThe Quantum-Mechanical Model: Atomswith Orbitals290291294Baseball Paths and Electron Probability Maps 294From Orbits to Orbitals 2959.6Quantum-Mechanical Orbitals and Electron Configurations295Quantum-Mechanical Orbitals 296Electron Configurations: How ElectronsOccupy Orbitals 298Sign of Hrxn 265Stoichiometry of Hrxn 266Chapter in reviewBlimps, Balloons, and Models of the AtomLight: Electromagnetic RadiationThe Electromagnetic SpectrumChemistry and Health Radiation9.79.8Electron Configurations and the Periodic Table 302The Explanatory Power of the Quantum- Mechanical Model30512/11/13 1:04 PM
Contents xi9.9Periodic Trends: Atomic Size, IonizationEnergy, and Metallic Character307Atomic Size 307Chemistry and Health Pumping Ions:Atomic Size and Nerve ImpulsesIonization Energy 309Metallic Character 311Chapter in reviewKey TermsExercises10ChemicalBonding30931331631632410.1 Bonding Models and AIDS Drugs32510.2 Representing Valence Electrons with Dots32610.3 Lewis Structures of Ionic Compounds: ElectronsTransferred10.4 Covalent Lewis Structures: Electrons Shared327328Double and Triple Bonds 32910.5 Writing Lewis Structures for CovalentCompounds330 Problem-Solving Procedure Writing LewisStructures for Covalent Compounds 331Writing Lewis Structures for Polyatomic Ions 332Exceptions to the Octet Rule 33310.6 Resonance: Equivalent Lewis Structuresfor the Same Molecule10.7 Predicting the Shapes of MoleculesChemistry in the Environment The Lewis Structure of Ozone11.3 Pressure: The Result of Constant334335336 Problem-Solving Procedure PredictingGeometry Using VSEPR Theory339Representing Molecular Geometries on Paper 339Chemistry and Health Fooled byMolecular Shape34010.8 Electronegativity and Polarity: Why Oiland Water Don’t MixElectronegativity 341Polar Bonds and Polar Molecules 343Everyday Chemistry How Soap WorksChapter in reviewKey TermsExercises11345346349349362Pressure Units 363Pressure Unit Conversion 36411.4 Boyle’s Law: Pressure and VolumeEveryday Chemistry Airplane CabinPressurization11.511.611.711.8Everyday Chemistry Extra-long SnorkelsCharles’s Law: Volume and TemperatureThe Combined Gas Law: Pressure, Volume,and TemperatureAvogadro’s Law: Volume and MolesThe Ideal Gas Law: Pressure, Volume,Temperature, and Moles365366370371375377379Molar Mass of a Gas from the Ideal Gas Law 38311.9 Mixtures of Gases: Why Deep-Sea DiversBreathe a Mixture of Helium and Oxygen385Deep-Sea Diving and Partial Pressure 387Collecting Gases over Water 38811.10 Gases in Chemical Reactions389Molar Volume at Standard Temperatureand Pressure 392Gases11.1 Extra-Long Straws11.2 Kinetic Molecular Theory: A Model for GasesTro IC5 FM i-xxxv v4.0.1.indd 11341Molecular CollisionsChemistry in the Environment 358Air PollutionChapter in review359360Key TermsExercises39439540040012/11/13 1:04 PM
xii ContentsDipole–Dipole Force 426Hydrogen Bonding 428429Ion–Dipole Force Chemistry and Health HydrogenBonding in DNA12.7 Types of Crystalline Solids: Molecular,Ionic, and Atomic430432Molecular Solids 432Ionic Solids 433Atomic Solids 43312.8 Water: A Remarkable MoleculeChemistry in the Environment Water PollutionChapter in reviewKey TermsExercises13Solutions13.1 Tragedy in Cameroon13.2 Solutions: Homogeneous Mixtures13.3 Solutions of Solids Dissolved in Water:How to Make Rock Candy434435436440440446447448449Solubility and Saturation 450Electrolyte Solutions: Dissolved Ionic Solids 451How Solubility Varies with Temperature 452Rock Candy 452Liquids, Solids,and IntermolecularForces1212.1 Interactions between Molecules12.2 Properties of Liquids and Solids12.3 Intermolecular Forces in Action: SurfaceTension and Viscosity13.4 Solutions of Gases in Water: How SodaPop Gets Its Fizz13.5 Specifying Solution Concentration:Mass Percent410411412413Surface Tension 414Viscosity 414Everyday Chemistry Why Are WaterDrops Spherical?12.4 Evaporation and Condensation415415Boiling 417Energetics of Evaporation and Condensation 418Heat of Vaporization 41912.5 Melting, Freezing, and Sublimation420Energetics of Melting and Freezing 421Heat of Fusion 421Sublimation 423Dipole–Dipole, Hydrogen Bonding, andIon–DipoleDispersion Force 425Tro IC5 FM i-xxxv v4.0.1.indd 1213.6 Specifying Solution Concentration: MolarityChemistry in the Environment The Dirty DozenUsing Molarity in Calculations 459Ion Concentrations 46113.7 Solution Dilution13.8 Solution Stoichiometry13.9 Freezing Point Depression and BoilingPoint Elevation: Making Water FreezeColder and Boil HotterFreezing Point Depression 466Everyday Chemistry Antifreeze in FrogsBoiling Point Elevation 46813.10 Osmosis: Why Drinking Salt Waterin MedicineChapter in review425454Mass Percent 454Using Mass Percent in Calculations 455Causes DehydrationChemistry and Health Solutions12.6 Types of Intermolecular Forces: Dispersion,452Key 11/13 1:05 PM
Contents xiii Calculating pH from [H3O ] 508Calculating [H3O ] from pH 509The pOH Scale 51014.10 Buffers: Solutions That Resist pH ChangeChemistry and Health AlkaloidsChemistry and Health The Danger ofAntifreezeChapter in reviewKey ium52815.1 Life: Controlled Disequilibrium15.2 The Rate of a Chemical Reaction52953015.3 The Idea of Dynamic Chemical Equilibrium15.4 The Equilibrium Constant: A Measure of534Collision Theory 530How Concentration Affects the Rate of a Reaction 532How Temperature Affects the Rate of a Reaction 533How Far a Reaction Goes1414.114.214.314.415.5 Heterogeneous Equilibria: The EquilibriumAcids and Bases486Sour Patch Kids and International Spy MoviesAcids: Properties and ExamplesBases: Properties and ExamplesMolecular Definitions of Acids and Bases487488489490The Arrhenius Definition 490The Brønsted–Lowry Definition 49114.5 Reactions of Acids and BasesNeutralization Reactions 493Acid Reactions 494Everyday Chemistry What Is inMy Antacid?Base Reactions 49614.6 Acid–Base Titration: A Way to Quantify theAmount of Acid or Base in a Solution14.7 Strong and Weak Acids and Bases493496496499Strong Acids 499Weak Acids 500Strong Bases 503Weak Bases 50314.8 Water: Acid and Base in One14.9 The pH and pOH Scales: Ways toExpress Acidity and BasicityTro IC5 FM i-xxxv v4.0.1.indd 13537Writing Equilibrium Constant Expressions forChemical Reactions 537The Significance of the Equilibrium Constant 538Expression for Reactions Involving a Solidor a Liquid15.6 Calculating and Using Equilibrium Constants540541Calculating Equilibrium Constants 541Using Equilibrium Constants in Calculations 54315.7 Disturbing a Reaction at Equilibrium:Le Châtelier’s Principle54415.8 The Effect of a Concentration Changeon Equilibrium54615.9 The Effect of a Volume Change on Equilibrium 548Chemistry and Health How aDeveloping Fetus Gets Oxygen from Its Mother55015.10 The Effect of a Temperature Changeon Equilibrium55115.11 The Solubility-Product Constant553Using Ksp to Determine Molar Solubility 554Everyday Chemistry Hard Water15.12 The Path of a Reaction and the Effectof a Catalyst555556How Activation Energies Affect Reaction Rates 556Catalysts Lower the Activation Energy 558Enzymes: Biological Catalysts 559504Chapter in reviewKey Terms507Exercises56056456412/11/13 1:05 PM
xiv Contents16Oxidation andReduction57416.1 The End of the Internal Combustion Engine?16.2 Ox
vii Contents Preface xviii 1 The Chemical World 2 1.1 Soda Pop Fizz 3 1.2 Chemicals Compose Ordinary Things 5 1.3 All Things Are Made of Atoms and Molecules 5 1.4 The Scientific Method: How Chemists Think 6 EvEryday ChEmistry Combustion and the Scientific Method 8 1.5 A Beginning Chemist: How to Succeed 8 CHAPTER IN REvIEw 9 KEy TERMS 10 ExERCISES 10 2 Measurement and
Pearson Education LTD. Pearson Education Australia PTY, Limited. Pearson Education Singapore, Pte. Ltd. Pearson Education North Asia, Ltd. Pearson Education Canada, Ltd. Pearson Educatión de Mexico, S.A. de C.V. Pearson Education—Japan Pearson Education Malaysia, Pte. Ltd. Library of Co
Pearson Education LTD. Pearson Education Australia PTY, Limited. Pearson Education Singapore, Pte. Ltd. Pearson Education North Asia, Ltd. Pearson Education Canada, Ltd. Pearson Educación de Mexico, S.A. de C.V. Pearson Education—Japan Pearson Education Malaysia, Pte. Ltd. The Libra
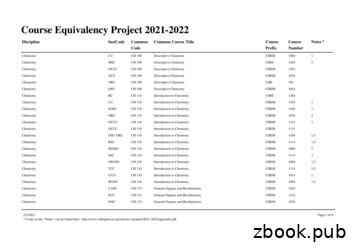
Chemistry ORU CH 210 Organic Chemistry I CHE 211 1,3 Chemistry OSU-OKC CH 210 Organic Chemistry I CHEM 2055 1,3,5 Chemistry OU CH 210 Organic Chemistry I CHEM 3064 1 Chemistry RCC CH 210 Organic Chemistry I CHEM 2115 1,3,5 Chemistry RSC CH 210 Organic Chemistry I CHEM 2103 1,3 Chemistry RSC CH 210 Organic Chemistry I CHEM 2112 1,3
Physical chemistry: Equilibria Physical chemistry: Reaction kinetics Inorganic chemistry: The Periodic Table: chemical periodicity Inorganic chemistry: Group 2 Inorganic chemistry: Group 17 Inorganic chemistry: An introduction to the chemistry of transition elements Inorganic chemistry: Nitrogen and sulfur Organic chemistry: Introductory topics
Pearson (UK) 80 Strand, London WC2R 0RL, UK T 44 (0)20 7010 2000 F 44 (0)20 7010 6060 firstname.lastname@pearson.com www.pearson.com Pearson (US) 1330 Avenue of the Americas, New York City, NY 10019, USA T 1 212 641 2400 F 1 212 641 2500 firstname.lastname@pearson-inc.com www.pearson.com Pearson Education One Lake Street, Upper Saddle River,
Pearson Education Canada, Inc. Pearson Education Malaysia, Pte. Ltd. Pearson Education-Japan Pearson Education Upper Saddle River, New Jersey Pearson Education Australia PTY, Limited PEARSON 10 9 8 7 6 5 4 ISBN-13: 17Ö-D-13-S0M507-7 ISBN-ID: G-13-5tmsa7-X . For Diane Perin Hock and Caroline Mei Perin Hock . CONTENTS PREFACE xi CHAPTER I BIOLOGY AND HUMAN BEHAVIOR 1 READING 1: ONE BRAIN OR TWO .
Accelerated Chemistry I and Accelerated Chemistry Lab I and Accelerated Chemistry II and Accelerated Chemistry Lab II (preferred sequence) CHEM 102 & CHEM 103 & CHEM 104 & CHEM 105 General Chemistry I and General Chemistry Lab I and General Chemistry II and General Chemistry Lab II (with advisor approval) Organic chemistry, select from: 9-10
CHEM 0350 Organic Chemistry 1 CHEM 0360 Organic Chemistry 1 CHEM 0500 Inorganic Chemistry 1 CHEM 1140 Physical Chemistry: Quantum Chemistry 1 1 . Chemistry at Brown equivalent or greater in scope and scale to work the studen