11 Chemistry - Question Bank For Inorganic Chemistry 1 .
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 995212256211th Chemistry - Question Bank for Inorganic Chemistry1. Chemical Calculations3 Marks (Book Inside)1. Define formula weight (FW) or formula mass.2. Define Avogadro’s number (NA).3. Define mole.4. Define molar mass.5. Define Empirical formula (or) simplest formula.6. What are the steps used to write the empirical formula.7. Define molecular formula.8. What are the steps to write the molecular formula?9. What is oxidation reaction?10. What is reduction reaction?11. Define oxidation in terms of electronic concept.12. Define reduction in terms of electronic concept.13. Define oxidation number (or) oxidation state.14. Define redox reaction.15. Define strength.16. Define Molarity.17. Define Normality.18. Define molality.19. Define mole fraction.20. Define the law of volumetric analysis.21. Write a note on determination of equivalent masses of elements by Hydrogendisplacement reaction.22. What are the uses of volumetric law?23. Write a note on equivalent mass of acid.24. Write a note on equivalent mass of base.25. Write a note on equivalent mass of salt.26. Write a note on equivalent mass of an oxidizing agent.27. Write a note on equivalent mass of an reducing study-materials-tamil-medium-english-medium.html
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 99521225625 Marks (Book Back)1. Can two different compounds have same molecular formula? Illustrate youranswer with two examples.2. What are the essentials of a chemical equation?3. What is the information’s conveyed by a chemical equation?4. Balance the following equations . i. Fe H2O Fe3O4 H2ii. Fe2 (SO4)3 NH3 H2O Fe(OH)3 (NH4)2 SO4iii. KMnO4 H2SO4 K2SO4 MnSO4 H2O O2iv. K2Cr2O7 H2SO4 K2SO4 Cr2(SO4)3 H2O O25 Marks (Book Inside)1. Explain the determination of molecular mass by Victor – Meyer’s method.2. Explain the determination of equivalent mass of elements by oxide method.3. Explain the determination of equivalent mass of elements by chloride method.4. Write a note on equivalent mass of oxidizing agent & reducing agent.5. Write a note on equivalent mass of the (a) acid (b) base (c) salt.6. Mention the rules for writing stoichiometric equation.7. What are the general rules for assigning oxidation number to an atom?8. Write the rules for balancing redox reaction by oxidation number method.2. General Introduction to Metallurgy3 . Marks (Book Back)1. Distinguish between ore and mineral with suitable example?2. What are the elements obtained from sea water source?3. What are the different methods of concentration of ores?4. What is gravity separation?5. Name the ores which are concentrated by froth floatation process.6. Define Metallurgy.7. What are the major steps involved in the metallurgical process?8. What is calcinations? Give example.9. What is the principle involved in Bessemer process?10. What is meant by electrolytic refining? Give example.11. What is anode mud?12. What do you understand by the following terms (i) roastin (ii) smelting(Book Inside) -----------------------------------13. Distinguish between ore and mineral with suitable example?14. What are the elements obtained from sea water source?15. What are the different methods of concentration of ores?16. What is gravity separation?17. Name the ores which are concentrated by froth floatation h-study-materials-tamil-medium-english-medium.html
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 995212256218. Define Metallurgy.19. What are the major steps involved in the metallurgical process?20. What is calcinations? Give example.21. What is the principle involved in Bessemer process?22. What is meant by electrolytic refining? Give example.23. What is anode mud?24. What do you understand by the following terms (i) roasting (ii) smelting?25. What is matrix (or) gangue?26. Define Minerals. Give one example.27. Define ores. Give one example.28. Give any six names of oxide ores with composition.29. Give any six names of sulphide ores with composition.30. Give any three names of carbonate ores with composition.31. Give any three names of halide ores with composition.32. Give any three names of sulphate ores with composition.33. Give any three names of silicate ores with composition.34. Write descending mass abundance of the elements in Humans.35. Write descending mass abundance of the elements in Oceans.36. Write descending mass abundance of the elements in Earth crust.37. What is acidic Bessemer process?38. What is Basic Bessemer process?39. Give any six metals which are purified by electrolytic refining.40. What are the metals purified by Zone refining method?41. Write a note on Mond’s process.5 Marks (Book Back)1. Write short note on source of element in living system.2. Explain froth flotation process with neat diagram.3. How electrolytic separation process is useful in the separation ofmagnetic impurities from nonmagnetic ores? Draw the diagram.4. How the impurities of ore are removed by chemical method?5. What is roasting? Explain different types of roasting with suitableexample.6. What is smelting? Explain the process with example.7. What is Zone refining? Describe the principle involved in thepurification of the metal by this method.8. How nickel is extracted by Mond’s process? Write the variousreactions involved in the process.9. Write short note on mineral wealth of India.10. Give a brief account of the mineral wealth of Tamil Nadu.(Book Inside) -----------------------------------11. Explain the Electrolytic refining of Copper.3. Atomic structure –I3 Marks (Book Back)1. What is the charge of an electron, proton and h-study-materials-tamil-medium-english-medium.html
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 99521225622.3.4.5.What is atomic number?What is the maximum number of electrons that an orbital can have?How many orbital’s are there in the second orbit? How are they designated?Sketch the shape of s and p-orbital indicating the angular distribution ofelectrons.6. What are the charge and mass of an electron?7. What is an orbital?8. Give the order of filling of electrons in the following orbital’s 3p, 3d, 4p,4sand6s.9. What is meant by principal quantum number?10. How many protons and neutrons are present in 8O18?11. What are the particles generally present in the nuclei of atoms?12. . Show how the atomof the element is constituted?13. How will you experimentally distinguish between a ray of neutron and ray ofproton?14. What is the principal defect of Bohr atom model?15. Write the complete symbol for(a)The nucleus with atomic ber26 andmassnumber55(c)The nucleus with atomic number 4 and mass number 9.16. An atomic orbital has n 3.Whatarethepossiblevaluesofl?17. An atomic orbital has l 3.Whatarethepossiblevaluesofm?18. Give the electronic configuration of chromium.(Z 24).19. Which energy level does not have p-orbital?20. erof p-orbital?21. How many electrons can haves ½inad-sub-shell?22. Write the values of l and m for p-orbital.23. Which quantum accounts for the orientation of the electron orbital?24. What is shape of the orbital with (i) n 2andl 0;(ii)n 2and l 1?25. Give the values forallquantumnumbersfor2pelectronsinnitrogen (Z 7).26. Give the electronic configuration of Mn2 and Cu. Atomic number ofCu 29andMn 25.27. Explain why the electronic configuration of Cr and Cu are written as 3d5,4s1 and3d10 4s1 instead of 3d4 4s2 and 3d9 4s2?(Book Inside) -----------------------------------28. Write a note on Thomson’s model of an atom.29. What is Rutherford’s alpha (α )-rays scattering experiment?30. Define atomic mass.31. Define Orbit.32. What is stationary state?33. State Heisenberg uncertainty principle.34. What is Zeeman Effect?35. What is Stark effect?36. Define Quantum -study-materials-tamil-medium-english-medium.html
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 995212256237. What are the types of Quantum number?38. Write a note on principal quantum number.39. Write a note on azimuthal (or) subsidiary quantum number.40. Write a note on Magnetic Quantum number.41. Write a note on Spin Quantum number.42. Write a note on shape of s – orbital.43. Write a note on shape of p – orbital.44. Write a note on shape of d – orbital.45. Define Pauli’s exclusion principle.46. Define Hund’s rule of maximum multiplicity.47.What is (n l ) rule.48.Write a note on stability of orbital.5 Marks (Book Back)1. Describe Aufbau principle .Explain its significance in the electronic buildup ofatoms.2. Using the s,p,d, notation, describe the orbital with the following quantumnumbers?(a)n 1,l 0;(b)n 2,l 0;(c)n 3,l 1; (d)n 4,l 3.3. Using the a Aufbau principle, write the electronic configuration in the groundstate of the following atoms : Boron(Z 5)Neon(Z 10) and Aluminium (Z 13).4. What is Rutherford’s alpha (α )- ray scattering experiment? What are itsconclusions?5. What are the postulates of Bohr Theory of an atom?6. Explain the various quantum numbers which completely specify the electron ofan atom.(Book -------------------------------------------7. Explain the defect of Rutherford’s model.8. Give any five limitations of Bohr’s model of an atom.9. Explain the shape of s, p – orbital.10. Explain the shape of d – orbital.11. Explain the Pauli’s exclusion principle with some examples.12. Explain the Hund’s rule of maximum multiplicity with some examples.4. Periodic classification – I3 Marks(Book Back)1. Arrange F,Cl, Br and I in the order of increasing electronic gain enthalpy.2. Write electronic configurations for the elements of atomic numbers 6 and14andfrom this find out of which group in the periodic table each elements belongs.3. Which of the following electronic configurations has the lowest )1s2,2s2, 2p6,3s2.4. State Modern Periodic Law.5. Why Noble gases have zero electron gain enthalpy?6. Which of the following pairs of elements would you expect to have lower firstionization enthalpy? (a)Cl or F; (b)Cl or S; (c)K or Ar; (d)Kr or dy-materials-tamil-medium-english-medium.html
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 99521225627. Why d o e l e m e n t s i n t h e same g r o u p h a v e g e n e r a l l y si m i l a rproperties?8. Name any two transition elements and any two inner transition elements.9. Arrange t h e order of increasing atomic volumes in: (a)Li, Na and K;(b)C,N and O;(c)Ca, Sr and Ba.10. Name the different blocks of elements in periodic table. Give the generalelectronic configuration of each block.11. belongs12. Why nitrogen has higher I.E. value than oxygen?13. Out o f f l u o r i n e a n d c h l o r i n e , w h i c h h a s g r e a t e r e l e c t r o n g a i nenthalpy?14. Why d-block elements are called transition elements?15. What property did Mendeleev use to classify elements in his periodic table?16. Among the elements Li,K,Ca, S and Kr which one has the lowest first ionizationenthalpy? Which has the highest first ionization enthalpy?(Book Inside) ----------------------------------17. Write a note on Dobereiner’s triads.18. Write a note on Newland law of octaves.19. Write a note on Lother Meyer’s arrangement of elements.20. Define Mendeleev’s periodic law.21. Write a note on s – block elements.22. What are representative (or) Main group elements?23. What are inner transition elements?24. Write the general electronic configuration of s,p,d,f, block elements.25. What are metalloids (or) semi metals?26. Arrange the following in the order of increasing metallic character.Be,Mg,Na,Si,P.27. Define atomic radii.28. Define ionic radii.29. Define Ionisation energy (or) Ionisation potential.30. Define iso electronic.31. Which of the following will have the largest and smallest size, Mg, Mg 2, Al, Al 3.32. What are the factors will affect the Ionisation energy.33. Define electron affinity (or) electron gain enthalpy.34. What are the factors will affect the electron affinity.35. Define electronegativity.36. Why Ionisation energy of B is less than that of Be.37. Why Ionisation energy of Be, Mg, N & P are higher.38. Why electron affinity of Be & Mg are zero.5 Marks (Book Back)1. Why does the first ionization enthalpy would have higher electron gain enthalpy?2. Which of the following pairs of elements would have higher electron gainenthalpy? (a) N or O ; (b) F or Cl . h-study-materials-tamil-medium-english-medium.html
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 99521225623. Lanthanides and actinides are placed in separate rows at the bottom of theperiodic table. Explain the reason for this arrangement?4. What do you mean by representative elements? Name the groups of the periodictable, which contain representative elements.5. Define transition elements. Name the different transition series.6. Which elements of the following pairs have smaller ionization enthalpy? (a) Ca orBe ; (b) Ca or K ; (c) Cl or I. Justify your answer.7. Why is Na atom bigger than the atoms of both lithium and magnesium ?8. What do you mean by the term electron gain enthalpy? How does electron gainenthalpy change along a period and in a group?9. Explain how the elements are arranged in the form of the periodic table.10. What are normal, transition and inner- transition elements?11. What are the differences between normal and transition elements?12. Explain why radii of positive ions are always smaller than the radii ofcorresponding neutral atoms and why negative ions have larger radii than thecorresponding neutral atom.13. Explain the size of group Cl- Na .14. What is electron gain enthalpy? On what factors does it depend?15. Give the general variation of electron gain enthalpies in the periodic table.16. Define the term ionic radius. Justify that the radius of anion is larger than theparent atom.17. What do you mean by ionization enthalpy? How does it vary across a period anddown a group?18. What is meant by electronegativity? On what factors does it depend?19. What are the essential features of the periodic table of Mendeleev?20. Discuss how his table has been modified subsequently.(Book Inside) ----------------------------------21. Explain the factors influencing Ionisation enthalpy (or) energy (or) potential.5. Group 1s – Block Elements.3 Marks (Book Back)1. What are isotopes? Mention the isotopes of hydrogen.2. Write a short note on tritium.3. How does deuterium react with nitrogen?4. How does deuterium react with metals?5. Mention the uses of deuterium.6. How is tritium prepared?7. How do you convert para hydrogen to ortho hydrogen?8. How does heavy water react with metals?9. How is hydrogen peroxide prepared in the laboratory?10. How is hydrogen peroxide solution concentrated?11. Write about the reducing property of hydrogen peroxide.12. Mention two important uses of H2O2.13. Why alkali metals have low melting and boiling -study-materials-tamil-medium-english-medium.html
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 995212256214. Why alkali metals have strong electropositive character?(Book Inside) ----------------------------------15. Write short note on protium (or) ordinary hydrogen.16. Write a note on Deuterium (or) heavy hydrogen.17. Write a note on preparation of Deuterium by electrolysis of heavy water.18. How does Deuterium react with oxygen & nitrogen?19. How does Deuterium react with halogens?20. Write any three exchange reaction of Deuterium.21. Mention the uses of Tritium.22. Write a note on ortho hydrogen.23. Write a note on para hydrogen.24. What is heavy water?25. How does heavy water react with metallic oxide?26. How does heavy water react with acid anhydride?27. How does heavy water react with metal carbide?28. Write a note on electrolysis of D2O.29. Give two examples for exchange reaction of heavy water.30. Write about the oxidizing properties of H2O2.31. How hydrogen is obtained from the other energy source.32. Give any three general characteristic of alkali metals.33. Why alkali metals show oxidation state of 1?34. Write a note on reducing properties of alkali metals.35. Mention any three uses of Lithium.36. How does sodium react with air?37. How does sodium react with H2O & CO2?38. How does sodium react with HCl & NH3?39. How does sodium react with Alumina & Silica?40. Mention any three uses of sodium.5 Marks (Book Back)1. How is deuterium obtained by diffusion process?2. Write about the exchange reactions of deuterium.3. What are ortho and para hydrogen? Explain.4. Differentiate between ortho and para hydrogen.5. Explain the preparation of heavy water.6. Compare water and heavy water.7. Explain the oxidizing property of hydrogen peroxide.8. Explain how liquid hydrogen can be used as a fuel.9. How is lithium extracted from its ore?(Book Inside) edium-english-medium.html
www.Padasalai.NetK N SUBRAMANIwww.TrbTnpsc.com9943009170 & 995212256210. How does Deuterium react with (a) O2 (b) N2 (c) halogens (d) Na (e) C2H411. Write a note on preparation of Tritium.12. How do you convert para hydrogen to ortho hydrogen?13. How does heavy water react with (a) metal (b)metallic oxide (c) metal carbide.14. How does heavy water react with (a) SO3 (b) P2O5 (c) NaOH (d) NH4Cl.15. Explain the laboratory preparation of H2O2 ?16. Mention any five general characteristics of alkali metals.17. Write a note on (a) Electropositive character (b) Oxidizing state(c) Reducing properties of alkali metals.18. Mention any five uses of Lithium.19. How does sodium react with (a) O2 (b) H2O (c)CO220. How does sodium react with (a) NH3 (b) HCl (c) Alumina (d) Silica.21. Mention any five uses of Sodium.6. Group 2s – Block Elements3 Marks (Book Back)1. Why the oxides of Group 2 metals have high melting points?3 2. Why there is increase in the ionisation potential for forming M ion for group 2metals?2 3. Why the ionization potential of M is not very much greater than M ?4. Why a precipitate of Mg(OH)2 is not formed when aqueous ammonia, NH4OH isadded to a solution of MgCl2?5. List the carbonates and hydroxide of alkaline earth metals in order of theirincreasing stability and their solution.6. Why do beryllium halides fume in air?7. Why group 2 elements are harder than alkali metals?8. Beryllium halides are covalent whereas magnesium halides are ionic.Why?9. Why monoxides of alkaline earth metals are are very stable?10. The basic strength of the oxides of group 2 elements increases from Be to Ba.Why?(Book Inside) ----------------------------------11. What are alkaline earth metals?12. Write the electronic configuration of Sr , Ba , Ra .13. Write a note on metallic properties of alkaline earth metals.14. Write a note on atomic radius of alkaline earth metals.15. Write a note on ionic radius of alkaline earth metals.16. Write a note on Ionization energy of alkaline earth metals.17. Write a note on Oxidation state of alkaline earth -study-materi
Nov 11, 2017 · 11th Chemistry - Question Bank for Inorganic Chemistry 1. Chemical Calculations 3 Marks (Book Inside) 1. Define formula weight (FW) or formula mass. 2. Define Avogadro’s number (N A). 3. Define mole. 4. Define molar mass. 5. Define Empirical formula (or) simplest formula. 6. W
Bruksanvisning för bilstereo . Bruksanvisning for bilstereo . Instrukcja obsługi samochodowego odtwarzacza stereo . Operating Instructions for Car Stereo . 610-104 . SV . Bruksanvisning i original
Northern Bank & Trust Co. Patriot Community Bank People's United Bank Pilgrim Bank Radius Bank RTN Federal Credit Union Santander StonehamBank TD Bank The Cooperative Bank The Savings Bank The Village Bank Walpole Cooperative Bank Wellesley Bank Winchester Co-operative Bank Abington Bank Bank of Canton Blue Hills Bank Boston Private Bank & Trust
M/s G.M. Kapadia & Co., Chartered Accountants Bankers HDFC Bank Ltd. (Primary Banker) Axis Bank Ltd. Bank of Baroda Bandhan Bank Ltd. Citibank N.A. CSB Bank Ltd. DCB Bank Ltd. Deutsche Bank ESAF Small Finance Bank ICICI Bank Ltd. IDFC Bank Ltd. Indian Bank RBL Bank Ltd. Saraswat Co-op Bank Ltd. State Bank of India Suryoday Small Finance Bank Ltd.
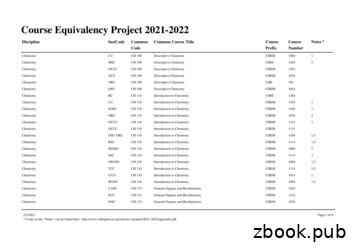
Chemistry ORU CH 210 Organic Chemistry I CHE 211 1,3 Chemistry OSU-OKC CH 210 Organic Chemistry I CHEM 2055 1,3,5 Chemistry OU CH 210 Organic Chemistry I CHEM 3064 1 Chemistry RCC CH 210 Organic Chemistry I CHEM 2115 1,3,5 Chemistry RSC CH 210 Organic Chemistry I CHEM 2103 1,3 Chemistry RSC CH 210 Organic Chemistry I CHEM 2112 1,3
10. HDFC Bank Limited 11. ICICI Bank Ltd 12. Indian Overseas Bank 13. ING Vysya Bank 14. Kotak Bank -Virtual card 15. Shivalik Bank 16. Standard Chartered Bank 17. State Bank of Bikaner and Jaipur 18. State Bank of India 19. State Bank of Mysore 20. State Bank of Travencore 21. Syndicate Bank 22. The Federal Bank Ltd 23. The Karur Vysya Bank Ltd
10 tips och tricks för att lyckas med ert sap-projekt 20 SAPSANYTT 2/2015 De flesta projektledare känner säkert till Cobb’s paradox. Martin Cobb verkade som CIO för sekretariatet för Treasury Board of Canada 1995 då han ställde frågan
service i Norge och Finland drivs inom ramen för ett enskilt företag (NRK. 1 och Yleisradio), fin ns det i Sverige tre: Ett för tv (Sveriges Television , SVT ), ett för radio (Sveriges Radio , SR ) och ett för utbildnings program (Sveriges Utbildningsradio, UR, vilket till följd av sin begränsade storlek inte återfinns bland de 25 största
Hotell För hotell anges de tre klasserna A/B, C och D. Det betyder att den "normala" standarden C är acceptabel men att motiven för en högre standard är starka. Ljudklass C motsvarar de tidigare normkraven för hotell, ljudklass A/B motsvarar kraven för moderna hotell med hög standard och ljudklass D kan användas vid