2019 Chemistry National 5 Finalised Marking Instructions
NationalQualifications20192019 ChemistryNational 5Finalised Marking Instructions Scottish Qualifications Authority 2019These marking instructions have been prepared by examination teams for use by SQA appointedmarkers when marking external course assessments.The information in this document may be reproduced in support of SQA qualifications only on a noncommercial basis. If it is reproduced, SQA must be clearly acknowledged as the source. If it is to bereproduced for any other purpose, written permission must be obtained from permission@sqa.org.uk.
General marking principles for National 5 ChemistryThis information is provided to help you understand the general principles you must apply whenmarking candidate responses to questions in this paper. These principles must be read inconjunction with the detailed marking instructions, which identify the key features required incandidate responses.(a)Marks for each candidate response must always be assigned in line with these generalmarking principles and the detailed marking instructions for this assessment.(b)If a specific candidate response does not seem to be covered by either the principles ordetailed marking instructions, and you are uncertain how to assess it, you must seekguidance from your team leader.(c)Marking should always be positive. This means that, for each candidate response, marksare accumulated for the demonstration of relevant skills, knowledge and understanding:they are not deducted from a maximum on the basis of errors or omissions.A guiding principle in marking is to give credit for correct chemistry rather than to look forreasons not to award marks.Example 1: The structure of a hydrocarbon found in petrol is shown below.Name the hydrocarbon.Although the punctuation is not correct, ‘3, methyl-hexane’ should gain the mark.Example 2: A student measured the pH of four carboxylic acids to find out how theirstrength is related to the number of chlorine atoms in the molecule.The results are shown in the table.Structural Cl3COOH0·51State how the strength of the acids is related to the number of chlorine atoms in themolecule.Although not completely correct, an answer such as ‘the more Cl2, the stronger the acid’should gain the mark.(d)There are no half marks awarded.(e)Candidates must respond to the ‘command’ word as appropriate and may be required towrite extended answers in order to communicate fully their knowledge and understanding.page 02
(f)Marks should be awarded for answers that have incorrect spelling or loose language as longas the meaning of the word(s) is conveyed. Example: Answers like ‘distilling’ (for‘distillation’) and ‘it gets hotter’ (for ‘the temperature rises’) should be accepted.However, the example below would not be given any credit, as an incorrect chemicalterm, which the candidate should know, has been given.Example: If the correct answer is ‘ethene’, and the candidate’s answer is ‘ethane’, thisshould not be accepted.(g)A correct answer followed by a wrong answer should be treated as a cancelling error andno marks should be awarded.Example: State what colour is seen when blue Fehling’s solution is warmed with analdehyde.The answer ‘red, green’ gains no marks.If a correct answer is followed by additional information which does not conflict, theadditional information should be ignored, whether correct or not.Example: State why the tube cannot be made of copper.If the correct answer is related to a low melting point, ‘Copper has a low melting pointand is coloured grey’ would not be treated as having a cancelling error.(h)Unless a numerical question specifically requires evidence of working to be shown, fullmarks should be awarded for a correct final answer (including units if required) on its own.The partial marks shown in the marking scheme are for use when working is given but thefinal answer is incorrect. An exception is when candidates are asked to ‘Find, bycalculation’, when full marks cannot be awarded for the correct answer without working.(i)In most questions units are not required. However, if the candidate writes units then theymust be correct. An incorrect unit would not be acceptable and one mark would not beawarded.This marking instruction must only be applied a maximum of once per paper.(j)Where the marking instructions specifically allocate a mark for units in a calculation, thismark should not be awarded if the units are incorrect or missing. Missing or incorrect unitsat intermediate stages in a calculation should be ignored.(k)As a general rule, where a wrong numerical answer (already penalised) is carried forwardto another step, credit will be given provided the result is used correctly. The exceptionto this rule is where the marking instructions for a numerical question assign separate‘concept marks’ and an ‘arithmetic mark’. In such situations, the marking instructions willgive clear guidance on the assignment of partial marks.(l)Ignore the omission of one H atom from a full structural formula provided the bond isshown or one carbon to hydrogen bond missing provided the hydrogen is shown.(m) A symbol or correct formula should be accepted in place of a name unless statedotherwise in the marking instructions.page 03
(n)When formulae of ionic compounds are given as answers it will only be necessary to showion charges if these have been specifically asked for. However, if ion charges are shown,they must be correct. If incorrect charges are shown, no marks should be awarded.(o)If an answer comes directly from the text of the question, no marks should be awarded.Example: A student found that 0·05 mol of propane, C3H8 burned to give 82·4 kJ ofenergy. Name the type of enthalpy change which the student measured.No marks should be awarded for 'burning' since the word ‘burned’ appears in the text.(p)Unless the question is clearly about a non-chemistry issue, eg costs in industrial chemicalprocess, a non-chemical answer gains no marks.Example: Suggest why the (catalytic) converter has a honeycomb structure.A response such as ‘to make it work’ may be correct but it is not a chemical answer andthe mark should not be awarded.page 04
Marking instructions for each B118.D119.C120.C121.C122.A123.D124.C125.B1page 05
Section 2Question1.(a)(b)Expected responseMaxmark(Covalent) Network1(i)Isotope(s)1(ii)Different number of neutrons1OROne atom has 5 neutrons and oneatom has 6 neutrons (both numbersmust be given)Additional guidanceAward zero marks for - covalent onits own.Award zero marks for-different mass in the nucleus-different number of particles in thenucleusORMore/less neutrons(iii) 11 /(c)11B/1115B120 (g)1Unit is not required however zeromarks are awarded for the correctvalue with an incorrect unit.This marking instruction must onlybe applied a maximum of once perpaper.2.(a)Hydroxyl1Award zero marks for – OH howeverthis does not negate the correctanswer.(b)Enzyme1Accept (Biological) Catalyst.(c)Calcium oxalate1(d)90 (g)1Unit is not required however zeromarks are awarded for the correctvalue with an incorrect unit.This marking instruction must onlybe applied a maximum of once perpaper.page 06
Question3.(a)Expected responseMust show all outer electronsMaxmark1Additional guidanceAccept cross or dot or e or e- torepresent electrons or a mixture ofthese.Accept petal diagram.Bonding electrons MUST be on theline or in the overlapping area.Element symbols do not need to beshown.If inner electrons are shown theymust be correct.(b)(c)(i)Blue/purple1Any colour that clearly represents analkaline pH is acceptable.As the temperature increasesyield/% decreases.1Award zero marks for wrong causeand effect eg as yield of ammoniadecreases, temperature gets higheretc.As the temperature decreases theyield/% increases.The yield/% increases as thetemperature decreases.The yield/% decreases as thetemperature increases.page 07
3.QuestionExpected responseMaxmark(c)For appropriate format: points (not bars)(1 mark)4(ii)The axis/axes of the graph has/havesuitable scale(s). For the graph paperprovided within the question paper, theselection of suitable scales will result ina graph that occupies at least half of thewidth and half of the height of the graphpaper(1 mark)Additional guidanceWhere the candidate has drawn abar graph the format mark is notawarded, but the remaining threemarks can still be accessed.For bar graphs, this mark is awardedfor the selection of a suitable scalefor percentage yield of ammoniaonly.The axes of the graph have suitablelabels and units(1 mark)Spelling mistakes or the use ofabbreviations should not bepenalised if the meaning of an axislabel may be clearly understood.All data points plotted accurately with aline of best fit (smooth curve/straight line) drawn.(1 mark)Where the candidate has drawn abar graph, the mark for accurateplotting can be awarded if theheights of bars are plottedaccurately but in this case no line ofbest fit should be used.If the scale is non-linear then themark for accurate plotting can onlybe accessed if the error occurs outwith the data-set.(d)(i)Electrolysis and lithium nitrideBoth required for1(1 mark)page 08
Question(ii)Expected responseArrow from water at top to water atbottom OR to the vertical line beneaththe ‘lithium nitride’ box.Maxmark1Additional guidanceDirection of arrow must be shown.ORArrow from lithium hydroxide at bottomto lithium hydroxide OR heat at top ORto the vertical line between lithiumhydroxide and heat.page 09Arrow to “molten lithium hydroxide”or the vertical line between “heat”and “molten lithium hydroxide” isawarded zero marks.
Question4.(a)(b)(i)Expected responseMaxmarkNucleus1The time for half of the mass/(radio)activity/nuclei to decay1Additional guidanceRadiation acceptable in place ofradioactivity.Any mention of reactivity – zeromarks.(ii)87·5% / 0·875 /Partial marking3 half-lives78(3 marks)3(1 mark)ORCorrect percentage or fraction leftfor a correct number of half-lives.(2 mark)ORCorrect percentage or fraction leftfor an incorrect number of halflives. Working must be shown.(1 mark)ORIf a candidate does not demonstratethe application of half-lives theycannot access the final mark forcalculating the percentage/fractiondecayed.Correct percentage or fractiondecayed with incorrect number ofhalf-lives. Working must be shown.(2 marks)eg824x 100 33·3%100 – 33·3 66·7%Zero marks.(iii) When an iodine-131 solution isdiluted,gets longerthe half-lifestays the samegets shorterpage 101
Question5.(a)Expected responseBromine/Br2 decolourisedMaxmark1ORAdditional guidanceAccept bromine/bromine water/bromine solution but do not acceptbromide or Br.Zero marks awarded for ‘goes clear’however if given in addition to acorrect answer it does not negate.bromine/Br2 goes colourlessIf starting colour is given it must becorrect eg orange/yellow/red-brownor brown.If candidate states correct answerfollowed by incorrect statementsuch as because it has a carbon tocarbon single bond zero marks ard zero marks for Cl or chloride1Accept polypropylene(iii) Polypropene/Poly(propene)page 11
5.QuestionExpected responseMaxmarkAdditional guidance(c)140 (g)(3 Partial marks3Rounding in intermediate steps doesnot negate the award of 3 marks butworking must be shown.(i)Both GFMsie 28 and 82(1 mark)ORMoles of cyclohexeneie (410 82) 5 mol(1 No units required but a maximum oftwo marks can be awarded if anincorrect unit is given. This markinginstruction must only be applied amaximum of once per paper.Award zero marks if thecandidate’s working does not usecyclohexene.A maximum of two marks can beawarded where the candidate hascarried out the calculation usingcyclohexene and butadieneprovided working is shown.1 concept mark for either:Candidate's GFM of ethene410 Candidate's GFM of cyclohexene(1 mark)ORMoles of cyclohexene Candidate’sGFM of ethene(1 mark)Where the candidate has beenawarded either concept mark, afurther mark can be awarded forcorrect follow through to a finalanswer(1 mark)page 12An incorrect GFM, with no workingshown, cannot be used to gain theconcept mark and thereforearithmetical follow through cannotbe accessed.
5.QuestionExpected responseMaxmarkAdditional guidance(c)(Cyclopentene) has smaller/weaker/less forces (of attraction) (1 mark)2The term bond is only acceptable ifit is specifically identified asbetween the molecules or used withthe term intermolecular.(ii)Between molecules or mention ofintermolecular attractions (1 mark)If neither of these two points aregiven, a maximum of 1 mark can beawarded for – cyclopentene issmaller/has fewer carbons orhydrogens or atoms/smaller carbonchainMention of breaking:- (more) bonds- bonds within molecule/chain- carbon to carbon bonds- carbon to hydrogen bondscannot gain the second mark butdoes not negate the first mark.Fewer bonds in the compound is notsufficient to imply a smallermolecule but does not negate.Candidates can be awarded thefull/partial marks if they explainwhy the cyclohexene has a higherboiling point. It must be clear fromthe candidate’s response that theyare referring to cyclohexene.page 13
Question6.Expected responseThis is an open ended question1 mark: The student hasdemonstrated a limitedunderstanding of the chemistryinvolved. The candidate has madesome statement(s) that is/arerelevant to the situation, showingthat at least a little of the chemistrywithin the context is understood.2 marks: The student hasdemonstrated a reasonableunderstanding of the chemistryinvolved. The student makes somestatement(s) that is/are relevant tothe situation, showing that thecontext is understood.3 marks: The maximum availablemark would be awarded to a studentwho has demonstrated a goodunderstanding of the chemistryinvolved. The student shows a goodcomprehension of the chemistry ofthe situation and has provided alogically correct answer to thequestion posed. This type ofresponse might include a statementof the principles involved, arelationship or an equation, and theapplication of these to respond tothe context. This does not mean theanswer has to be what might betermed an “excellent” answer or a“complete” one.page 14Maxmark3Additional guidance
7.QuestionExpected responseMaxmark(a)They have the same general formula1ANDsimilar/same chemical propertiesBoth required for (1 mark)(b)(c)Additional guidanceAward zero marks for- molecular formula- structural formula- chemical formulaAward zero marks for- physical properties in placeof chemical propertieshowever, it does not negate ifgiven in addition to chemicalproperties.(i)Alkane1(ii)C35 H721Numbers must be smaller thansymbol or subscriptSymbols must be correctZero marks awarded for:C35 H72c35 H72C35 h72c35 h72Accept 391 to 394 inclusive.1Unit is not required; however zeromarks are awarded for the correctvalue with an incorrect unit.This marking instruction must onlybe applied a maximum of once perpaper.8.(a)Supernova (explosions)1(b)Beryllium, aluminium, silicon andoxygen Be/Al/Si/O1Accept O2 and loose spelling ofelement names.1Equation does not need to bebalanced but if balanced it must becorrect:BeCl2 2K Be 2KClAll 4 required for (1 mark)(c)BeCl2 KBe KCl(d)Reduction1Redox not acceptable(e)10n1Do not accept1110N, n neutronpage 15
Question9.(a)(b)(i)Expected responseMaxmarkAdditional guidanceExothermic/exothermal1Do not accept combustion.0·05 (kg)(3 Partial markingUsing the correct concept of3No units required but a maximum oftwo marks can be awarded if anincorrect unit is given. This markinginstruction must only be applied amaximum of once per paper.m Ehwithc T8360 and 4180 can be used totogether in the calculation.c 4·18(1 mark)(To be awarded this concept markcandidates do not have tospecifically write m For values8·36 and 40Eh.)c T8360 and 40 can be awarded onemark provided c 4180.(1 mark)A further mark can be awarded forarithmetical follow through to thecandidate’s answer only if the markfor the concept has been awarded.(1 mark)(ii)Copper can: copper is a betterconductor (of heat) (than glass)(1 mark)2These two marks are independent ofeach other.A comparison of energy transfer tothe water must be made howeverzero marks are awarded for acomparison in terms of easier/quicker/faster.Heat shield: prevent/less heat loss(to the surroundings) or insulate theexperiment.(1 mark)(c)(i)If the OH/functional group is at theend/start/carbon number 1 (of thealcohol), an aldehyde is produced.Link must be made to heat/energy.1ORIf the OH/functional group is not atthe end/in the middle (of analcohol), a ketone is produced.(ii)1page 16
Question10.(a)(b)Expected responseMaxmarkIon bridge/salt bridge1(i)From right to left either on the wireor nearer the wire/voltmeter thanthe ion-bridge.1(ii)Oxidation1(iii) 2Fe3 2I 2Fe2 I21Additional guidanceZero marks awarded if arrow goes into solution arrow is drawn on wireand ion bridge arrow is drawn closer tothe ion bridge than thewire/voltmeterAccept correct multiples.Zero marks awarded for-electrons shown in the equation,unless clearly scored out.State symbols are not required andshould be ignored.(c)Conducts (electricity)1Additional correct properties do notnegate.Accept a correct description ofelectrical conductivity eg electronsfree to move or has delocalisedelectrons.page 17
Question11.Expected responseMaxmarkAdditional guidance(a)Ca2 CO32-1(b)Propanoic acid.1Accept propaneoic or propionic acid.(c)until 1Award zero marks for- any mention of dissolving- saturated solutionno more solid reacts/until it nolonger reactssolid remains/is left (at the bottomof the beaker)a gas is no longer produced no morefizzing/bubblingno more calcium carbonate reacts/calcium carbonate left (at thebottom)neutral/neutralised with adescription of testing pH.(d)neutral/neutralised on its own – zeromarks.1st technique - Filtration12nd technique - EvaporationAccept a correct description offiltration.Do not accept ‘use filter paper’ onits own as a description of filtration.Accept heat it/warm it/boil it/leaveby a window/leave for some time/distillation.Both required in correct order forone mark.page 18The term ‘dry’ for the 2nd techniqueis awarded zero marks but does notnegate a correct answer.
Question12.(a)Expected response(i)Standard (solution)(ii)21·2 or 21 (g)MaxmarkAdditional guidance1(3 marks)3Partial Marks can be awarded for amaximum of two of the followingthree steps:No units required but a maximum oftwo marks can be awarded if anincorrect unit is given. This markinginstruction must only be applied amaximum of once per paper.1 mark for correctly calculating thenumber of moles of sodiumcarbonate.ie n CV 1 x 0·2 0·2 mol(1 mark)1 mark for correctly calculating theGFM of sodium carbonateie GFM 106(1 mark)1 mark for calculating the mass ofsodium carbonateAn incorrect number of moles orGFM of sodium carbonate must besupported by working.ie m n x GFM usingcandidates calculated moles ofsodium carbonate and candidatescalculated GFM(1 mark)(b)(i)Any from:1Move burette/sodium carbonate(reading) to eye level.Reduce distance between flask andburetteUse white tile/paperClamp the burette towards thecentre.(ii)To show the end-point/point ofneutralisation/completion of thereaction(iii) Concordant11page 19Award zero marks for - repeat theexperiment
Question12.(b)(iv)Expected response1·5 (mol l-1)(3 Partial marks can be awarded for amaximum of two of the followingthree steps:Method A1 mark for the correct number ofmoles of sodium carbonate.ie n CV 1 0·015 0·015 mol(1 mark)1 mark for calculating the moles ofhydrochloric acid by correctlyapplying the molar ratioie 2 x candidate’s calculated numberof moles of Na2CO3(1 mark)0·03 mol HCl on its ownMaxmarkAdditional guidance3No units required but a maximum oftwo marks can be awarded if anincorrect unit is given. This markinginstruction must only be applied amaximum of once per paper.Accept mol l-1 or mol/l but notmol/l-1 or mol-1 or mol lAward zero marks if thecandidate’s working makes it clearthat they have not used sodiumcarbonate.If candidate does not attempt tocalculate the number of moles ofNa2CO3 then partial marks can beawarded for applying the molar ratiousing 1 mol of sodium carbonaterequires 2 moles of hydrochloric acidand/or using 2 moles of hydrochloricacid and V 0·02.ie 1:2 (1 mark)2/0·02 100 (1 mark)(2 marks)1 mark for calculating theconcentration of the hydrochloricacid.ie C n/V usingcandidate’s calculated number ofmoles of HCl and 0·02(1 ethod BC1 20 1 1521(1 mark)(1 mark)C1 20 30 or C1 10 15(this step on its own gets 2 marks)For method B using relationshipshown in the data book1 mark is awarded for the correctpairings of volume (in the same unit)and concentration.1 mark is awarded for the correctmole ratio being applied.OrC1 0·02 1 0·01521(1 mark)(1 mark)C1x 0·02 0·03(this step on its own gets 2 marks)page 201 mark is awarded for the correctarithmetic. This mark can only beawarded if an appropriate methodhas been used.
Question13.Expected responseMaxmark1 mark: The student hasdemonstrated a limitedunderstanding of the chemistryinvolved. The candidate has madesome statement(s) that is/arerelevant to the situation, showingthat at least a little of the chemistrywithin the context is understood.32 marks: The student hasdemonstrated a reasonableunderstanding of the chemistryinvolved. The student makes somestatement(s) that is/are relevant tothe situation, showing that thecontext is understood.3 marks: The maximum availablemark would be awarded to a studentwho has demonstrated a goodunderstanding of the chemistryinvolved. The student shows a goodcomprehension of the chemistry ofthe situation and has provided alogically correct answer to thequestion posed. This type ofresponse might include a statementof the principles involved, arelationship or an equation, and theapplication of these to respond tothe context. This does not mean theanswer has to be what might betermed an “excellent” answer or a“complete” one.[END OF MARKING INSTRUCTIONS]page 21Additional guidance
2019 2019 Chemistry National 5 Finalised Marking Instructions Scottish Qualifications Authority 2019 These marking instructions have been prepared by examination teams for use by SQA appointed markers when marking external course assessments. The information in this document may be r
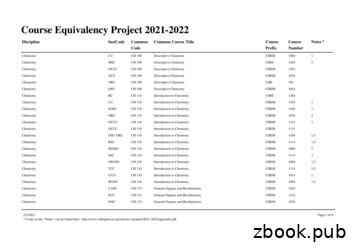
Chemistry ORU CH 210 Organic Chemistry I CHE 211 1,3 Chemistry OSU-OKC CH 210 Organic Chemistry I CHEM 2055 1,3,5 Chemistry OU CH 210 Organic Chemistry I CHEM 3064 1 Chemistry RCC CH 210 Organic Chemistry I CHEM 2115 1,3,5 Chemistry RSC CH 210 Organic Chemistry I CHEM 2103 1,3 Chemistry RSC CH 210 Organic Chemistry I CHEM 2112 1,3
Physical chemistry: Equilibria Physical chemistry: Reaction kinetics Inorganic chemistry: The Periodic Table: chemical periodicity Inorganic chemistry: Group 2 Inorganic chemistry: Group 17 Inorganic chemistry: An introduction to the chemistry of transition elements Inorganic chemistry: Nitrogen and sulfur Organic chemistry: Introductory topics
EMIR Update – ESMA Publishes Finalised Technical Standards – 27 September 2012 1 October 2012 EMIR Update - ESMA Publishes Finalised Technical Standards Introduction The European Securities and Markets Authority (“ESMA”) published on 27 September its technical standards and final report on EMIR, 4 days before
Accelerated Chemistry I and Accelerated Chemistry Lab I and Accelerated Chemistry II and Accelerated Chemistry Lab II (preferred sequence) CHEM 102 & CHEM 103 & CHEM 104 & CHEM 105 General Chemistry I and General Chemistry Lab I and General Chemistry II and General Chemistry Lab II (with advisor approval) Organic chemistry, select from: 9-10
CHEM 0350 Organic Chemistry 1 CHEM 0360 Organic Chemistry 1 CHEM 0500 Inorganic Chemistry 1 CHEM 1140 Physical Chemistry: Quantum Chemistry 1 1 . Chemistry at Brown equivalent or greater in scope and scale to work the studen
2019 2019 Physics National 5 Finalised Marking Instructions . marking candidate responses to questions in this paper. These principles must be read in conjunction . Question Answer Mark 1. A 1 2. B 1 3. E 1 4. C 1 5. C 1 6. B 1 7. C 1
2019 Alfa Romeo Giulia 2019 BMW X7 2019 Alfa Romeo Stelvio 2019 BMW Z4 2019 Audi A3 2019 Buick Cascada 2019 Audi A4 2019 Buick Enclave 2019 Audi A5 2019 Buick Encore 2019 Audi A6 2019 Buick Envision 2019 Audi A7 2019 Buick LaCrosse 2019 Audi A8 2019 Buick Regal 2019 Audi Allroad
“Am I My Brother’s Keeper?” Cain & Abel by Tintoretto. Everything can be taken from a man but the last of the human freedoms - to choose one’s attitude in an given set of circumstances, to choose one’s own way.--Auschwitz Survivor, Victor E. Frankl Human Gene Map. OnegShabbat Archives –Emanuel Ringleblum Remembrance: To record and to teach future Generations. The time has come to .