Medicinal Chemistry/ CHEM 458/658 Chapter 3- SAR And
Medicinal Chemistry/ CHEM458/658Chapter 3- SAR and QSARBela TorokDepartment of ChemistryUniversity of Massachusetts BostonBoston, MA1
Introduction Structure-Activity Relationship (SAR)- similar structures –similar effects- more potency or improved side effects Quantitative Structure-Activity Relationship (QSAR)- similar structures –similar effects but uses parameters todescribe the potency- parameters – anything (related to drug action) that can berepresented by a numerical values2
Structure-Activity Relationship (SAR) Usually go through minor changes on the lead structure- the and shape of the carbon skeleton- the nature and degree of substitution- stereochemistry3
Structure-Activity Relationship (SAR) Changing size and shape- number of methylene groups in chains and rings- increasing or decreasing the degree of unsaturation- introducing or removing a ring system4
Structure-Activity Relationship (SAR) Changing the number of methylene groups- increases lipophilicity (increased activity)- decreases water solubility (decreased activity)- aliphatic compounds – micelle formation – no selective bindingantipsychoticantidepressant5
Structure-Activity Relationship (SAR) Changing the degree of unsaturation- increasing – rigidity- E-Z isomers might complicate the picture- more sensitivity- increased toxicity1:30antipsychoticantidepressant6
Structure-Activity Relationship (SAR) Introduction or removal of a ring system- addition – size increase, shape changes (effect mostlyunpredictable- increasing size – better fills the hydrophobic pocket- small ring to substitute C C double bonds - stabilityantidepressant7
Structure-Activity Relationship (SAR) Introduction of an aromatic ring- increases rigidity, shape changes resistance toward metabolismmight improve8
Structure-Activity Relationship (SAR) Modifying the ring system of drugs of natural origin- fine tuning of effect and side effectsmore potent(highly addictive)less potent(less addictive)less potentequally potent(less addictive)9
Structure-Activity Relationship (SAR) Introduction of new substituentsmethyl groupsincreases olueneacetamideCH3 CONH 283ureaNH2 CONH 215propionamideN-methylureaStructureP490CH3 CH 2 CONH 2360CH 3NHCONH 244steric hindrance – might block activity013.710
Structure-Activity Relationship (SAR) Introduction of methyl group- methyl group on aromatic rings – increased rate of metabolism- demethylation – easy on heteroatoms, especially on N , S - reduce the rate of metabolism- reduce unwanted side effects11
Structure-Activity Relationship (SAR) Introduction of halogensMostly F and Cl - C-X bond stability - reactivityOH3CCF3 is also very popularCF3OOHOFONHCH3Olocation12
Structure-Activity Relationship (SAR) Introduction of hydroxyl groupsMostly to increase hydrophilic characterPhenolic OH is special13
Structure-Activity Relationship (SAR) Introduction of basic groupsMostly to increase binding via H-bonding/acid baseinteractions14
Structure-Activity Relationship (SAR) Introduction of COOH and SO3H groupsMostly to increase binding via H-bonding/acid baseinteractions – in vivo salt formationintroduction to small leads – usually changes the activitySO3H – no significant effect except faster excretionOther S groups are rare - metabolism15
Structure-Activity Relationship (SAR) Changes the existing substituents of a leadisosteres - bioisosteres16
Structure-Activity Relationship (SAR) Changes the existing substituents of a leadisosteres - bioisosteres17
Structure-Activity Relationship (SAR) Case Study: SAR investigation to discover potent geminalbisphosphonatesfirst generationsecond generation18
Structure-Activity Relationship (SAR) Case Study: SAR investigation to discover potent geminalbisphosphonates19
Structure-Activity Relationship (SAR) Case Study: SAR investigation to discover potent geminalbisphosphonates20
Structure-Activity Relationship (SAR) Case Study:SAR investigationto discover potent geminalbisphosphonates21
Structure-Activity Relationship (SAR) Case Study: SAR investigation to discover potent geminalbisphosphonates22
Quantitative Structure-Activity Relationship(QSAR) QSAR – mathematical relationship (equations)biological effect vs. physicochemical parameters- lipophilicity- electron distribution- shape- size- partition coefficients- Hammett or Tafts constantsBiological activity F {parameters (s)}23
Quantitative Structure-Activity Relationship(QSAR) Regression Analysis24
Quantitative Structure-Activity Relationship(QSAR) Lipophilic parametersPartition coefficientlog (1/C) k1 log P k2(1) Toxicity of alcohols to red spiders:log (1/C) 0.69 log P 0.16r 0.979, n 14, s 0.087(2) The binding of misc. neutral molecules to bovine serum:log (1/C) 0.75 log P 2.30r 0.96, n 42, s 0.159(3) The binding of misc. neutral molecules to haemoglobin:log (1/C) 0.71 log P 1.51r 0.95, n 17, s 0.16(4) Inhibition of phenols on the conversion of P-450 to P-420 cytochromes:log (1/C) 0.57 log P 036r 0.979, n 13, s 0.13225
Quantitative Structure-Activity Relationship(QSAR) Lipophilic parametersPartition coefficient – often paraboliclog (1/C) -k1 (log P)2 k2log P k326
Quantitative Structure-Activity Relationship(QSAR) Lipophilic parametershypnosis (mice) with barbiturateslog (1/C) - 0.44 (log P)2 1.58 log P 1.93 (r 0.969)Hansch – logP 2 hypnotic (CNS drug)27
Quantitative Structure-Activity Relationship(QSAR) Lipophilic parameterslipophilic substituent constants (π) (or hydrophobic)contribution of substituents to Pπ logPX – logPHπ logP(C6H5Cl) – logP(C6H6) 2.84 – 2.13 0.71π π (substituent 1) π (substituent 2) . π (substituent n)28
Quantitative Structure-Activity Relationship(QSAR) Lipophilic parameterslipophilic substituent constants (π)Substituent X-H- CH 3-F- Cl- OH- NH 2- NO 2- OCH3XAliphatic systems R-X0.000.50- 0.170.39- 1.160.470.000.560.140.71- 0.67- 1.23- 0.28- 0.02O2 NXHO0.000.520.540.11- 0.46- 0.390.18X0.000.490.310.93- 0.87- 1.630.50- 0.12log (1/C) vs π high r and low s – important contributor29
Quantitative Structure-Activity Relationship(QSAR) Lipophilic parametersdistribution coefficients (D)ionizationD [ HAorganic ][ H aqueous ] [ A-aqueous ]for acidslog (P/D-1) pH -pK afor baseslog (P/D-1) pK a - pH30
Quantitative Structure-Activity Relationship(QSAR) Electronic parametersThe Hammett constant (σ)σx log K xKσ x log K x - log Kσ x pK - pK x31
Quantitative Structure-Activity Relationship(QSAR) Electronic parametersThe Hammett constant (σ)log (1/C) 2.282 σ - 0.34832
Quantitative Structure-Activity Relationship(QSAR) Steric parametersThe Taft steric parameter (Es)k (XCH2COOCH3)Es logk (CH3COOCH3) k (XCH2COOCH3) - k (CH3COOCH3)log BR 0.440Es - 2.204 (n 30; s 0.37; r 0.886)33
Quantitative Structure-Activity Relationship(QSAR) Steric parametersMolar refractivity (MR)MR (n2 - 1) M(n2 2) ρadditive – functional groups34
Quantitative Structure-Activity Relationship(QSAR) Hansch analysisdrug activity vs. measurable chemical propertiesmultiparameter approachtwo stages: - transport to the site of action- binding to the target sitelog 1/C k 1 (partition parameter) k 2(electronic parameter) k 3 (steric parameter) k 4log 1/C k 1 P - k 2P2 k 3σ k 4 Es k 5
Quantitative Structure-Activity Relationship(QSAR) Hansch analysisAccuracy :- Greater number of analogs – n 5x (x number of parameters)- biological data- the choice of parametersUse:- Asses the factors controlling the activity- predict optimum activity (ideal parameter values)Sources of parameters- CRC, CAS, Merck Index, etc.
Quantitative Structure-Activity Relationship(QSAR) Craig plotsUse with Hansch analysis:log 1/C 2.67π – 2.56σ 3.92
Medicinal Chemistry/ CHEM 458/658 Chapter 3- SAR and QSAR Bela Torok Department of Chemistry University of Massachusetts Boston Boston, MA 1. Introduction Structure-Activity Relationship (SAR) - similar structures –
Wowdzia Mark & Elan Box2l5l 658-2345 Wowdzia Mark Fax Line 658-2347 Wowdzia Tom 658-2254 Wyllie Eileen 658-2248 Vakimetz Steve Box3 68Vegreville 658-2423 Yakimetz Terry 658-2252 Yakimowich David Vegreville 658-2167 Vakimowich Ken 658-2363 Yakimowich Ken Faxline 658-2555 Yakimyshyn M
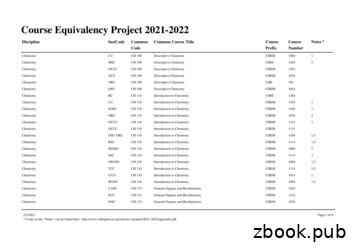
Chemistry ORU CH 210 Organic Chemistry I CHE 211 1,3 Chemistry OSU-OKC CH 210 Organic Chemistry I CHEM 2055 1,3,5 Chemistry OU CH 210 Organic Chemistry I CHEM 3064 1 Chemistry RCC CH 210 Organic Chemistry I CHEM 2115 1,3,5 Chemistry RSC CH 210 Organic Chemistry I CHEM 2103 1,3 Chemistry RSC CH 210 Organic Chemistry I CHEM 2112 1,3
Medicinal Chemistry/ CHEM 458/658 Chapter 2- Drug Structure and Solubility Bela Torok Department of Chemistry University of Massachusetts Boston Boston, MA 1. Structure Overall chemical structure - possible binding groups-size - sh
CHEM 350B Topics in Chemistry 7.5 454.95 CHEM 351 Chemicals Big and Small: Nano- 15 909.90 CHEM 352 Advanced Concepts in Chemistry 15 909.90 CHEM 352A Advanced Concepts in Chemistry 7.5 454.95 CHEM 352B Advanced Concepts in Chemistry 7.5 454.95 CHEM 360 Contemporary Green Chemistry 15 909.90 CHEM 380 Materials Chemistry 15 909.90
Accelerated Chemistry I and Accelerated Chemistry Lab I and Accelerated Chemistry II and Accelerated Chemistry Lab II (preferred sequence) CHEM 102 & CHEM 103 & CHEM 104 & CHEM 105 General Chemistry I and General Chemistry Lab I and General Chemistry II and General Chemistry Lab II (with advisor approval) Organic chemistry, select from: 9-10
CHEM 0350 Organic Chemistry 1 CHEM 0360 Organic Chemistry 1 CHEM 0500 Inorganic Chemistry 1 CHEM 1140 Physical Chemistry: Quantum Chemistry 1 1 . Chemistry at Brown equivalent or greater in scope and scale to work the studen
CHEM 31X. Chemical Principles 4 CHEM 33. Structure and Reactivity 4 CHEM 35. Organic Monofunctional Compounds 4 CHEM 36. Organic Chemistry Laboratory I 3 MATH 41, 42, 51. Calculus, Linear Equations 5 5 5 SECOND YEAR CHEM 130. Organic Chemistry Laboratory II 4 CHEM 131. Organic Polyfunctional Compounds y3 CHEM 134.
Description Logic RWTH Aachen Germany 4. Introduction to DL I A Description Logic - mainly characterised by a set of constructors that allow to build complex concepts and roles from atomic ones, concepts correspond to classes / are interpreted as sets of objects, roles correspond to relations / are interpreted as binary relations on objects, Example: Happy Father in the DL ALC Manu (9has-child .